2.1 Vaccine safety and approvals Te haumaru me ngā whakaaetanga o ngā kano ārai mate
Vaccine safety and approvals | Te haumaru me ngā whakaaetanga o ngā kano ārai mate
2.1.1 Introduction | Kupu whakataki
Vaccines are medicines used to protect the body against viruses and other infectious agents through immunisation. A vaccine trains the human immune system to respond as if it had been exposed to the virus or disease.118
Immunisation is a highly successful public health strategy. The World Health Organization estimates that every year immunisation prevents 3.5–5 million people from dying of diseases like diphtheria, tetanus, pertussis (whooping cough), influenza and measles.119
Vaccines are valuable in combating a pandemic, because they can prevent people from getting sick from a disease or reduce the severity of symptoms. Vaccination can reduce transmission of a disease and so promote 'herd immunity' (benefitting the vaccinated and unvaccinated alike).120 Widespread vaccination can relieve the burden on healthcare providers and delay changes (mutations) in the virus.121
2.1.1.1 Monitoring the safety of vaccines | Te aroturuki i te haumaru o ngā kano ārai mate
Many governments regulate vaccines as a type of medicine. In New Zealand, the regulator is Medsafe. Medsafe is a business unit in the Ministry of Health and is responsible for administering the Medicines Act 1981 and the Medicines Regulations 1984. This includes making decisions to approve medicines where their benefits outweigh their risks, and monitoring pharmacovigilance (post-marketing surveillance). Medsafe is a member of the International Coalition of Medicines Regulatory Authorities.
Under the Medicines Act 1981, Medsafe assesses safety of medicines in two key stages – through pre-market regulatory approval (see section 2.1.2 below) and during post-marketing surveillance (see section 2.1.6 below).122
Pharmacovigilance, defined as the science and activities relating to the detection, assessment, understanding and prevention of adverse events or medicines-related problems, aims to identify a 'safety signal'.123 This is information that raises the possibility of an adverse reaction to a medicine that requires further investigation.124 A wide range of sources, including reports of adverse reactions, literature and clinical studies can produce safety signals.125 New Zealand has had an established, government-led pharmacovigilance system since 1965.126
2.1.1.2 COVID-19 vaccine types and development | Ngā momo kano ārai mate KOWHEORI-19 me te whanaketanga
Because COVID-19 was a novel virus, there were no specific treatments for it in the early stages of the pandemic.127 International efforts to develop safe and effective vaccines moved at unprecedented speed and by July 2020 there were more than 170 COVID-19 vaccine development programmes underway.128 New Zealand ultimately used four COVID-19 vaccines: the Pfizer/BioNTech vaccine (Comirnaty), the AstraZeneca vaccine (Vaxzevria), the Janssen vaccine (Janssen), and the Novavax vaccine (Nuvaxovid).129
Comirnaty is an mRNA vaccine, using a technology that has been around for more than 20 years.130 The mRNA contained in these vaccines causes the human body to build copies of the distinctive 'spike' proteins found on the surface of the SARS-CoV-2 virus (not the whole virus). If the vaccinated person is later exposed to SARS-CoV-2, their immune system recognises the spike proteins as foreign and attacks the virus.131 Appendix 1 provides more detail on how mRNA vaccines work.
The Vaxzevria and Janssen vaccines are viral vector vaccines. They use a modified version of a virus (the 'vector') to deliver genetic instructions to the body's cells to make the spike protein. Again, because the vaccinated person's immune system has learnt to recognise the spike protein as foreign, it can defend against illness if it encounters the SARS-CoV-2 virus.132
Lastly, the Nuvaxovid vaccine uses artificially produced fragments of SARS-CoV-2 spike proteins assembled into nanoparticles, which cannot cause disease. The nanoparticles help a person's immune system to create antibodies and then to fight the SARS-CoV-2 virus if the person is infected.133
Standard vaccine development is a long process and the usual stages are:134
- testing the pharmaceutical quality using small batches. This determines an optimal formulation and manufacturing process that can keep vaccine components stable to the end of their shelf life, including in large-scale manufacture
- non-clinical laboratory studies on cells (in vitro)135 or on animals (in vivo)136
- three phases of clinical trials to evaluate safety and efficacy:
- Phase I trials generally involve between 20 to 100 participants and aim to establish if the vaccine triggers the expected immune response, if larger studies of the vaccine will be safe, and which doses are adequate.
- Phase II trials involve several hundred participants, and aim to establish the best doses to use, how many doses to use, and the most common side effects.
- Phase III trials include thousands of participants and help to establish clinical efficacy and safety, and to identify less common side effects.
The final stages of vaccine development involve scientific evaluation and authorisation, large-scale production, and studies following approval.
2.1.1.3 An expedited development of vaccines during the COVID-19 pandemic | Te whakawhanaketanga whakatere o ngā kano ārai mate i te wā o te urutā KOWHEORI-19
Some submitters were concerned that the development (and use) of COVID-19 vaccines was rushed, and important safeguards were abandoned (see discussion in Appendix 1). It is true that early in the pandemic, governments, regulators, manufacturers, ethical review bodies, scientists and doctors came together in pursuit of rapidly developing a safe and effective vaccine. However, it helped that research on mRNA vaccines and on improving vector-based vaccines was underway before the pandemic.137 With the early publication of the SARS-CoV-2 genomic sequence in January 2020, this enabled very quick development of new vaccines.138
The mRNA-based vaccines are faster to develop than other, conventional vaccine processes. Conventional vaccines rely on modifying and growing versions of the virus in processes that can take several months. In contrast, manufacture of mRNA-based vaccines uses a cell-free in vitro transcription process, which does not require culture of the virus. The process allows easy and rapid production within weeks, and the prospect of producing high numbers of vaccine doses within a short time.139
Pharmaceutical companies invested heavily in developing manufacturing processes in parallel with clinical trials, so they could scale up production quickly. They ran the three phases of clinical trials concurrently with the help of thousands of willing volunteers. A process that normally takes many years took less than a year, without missing any of the usual steps.140 People involved in developing COVID-19 vaccines told us that the primary focus was ‘maintaining the rigour while working at pace’.141
2.1.1.4 The Government's COVID-19 Vaccine Strategy aimed to provide a way out of restrictive public health measures and bring the pandemic to an end | Ko te whai o te Rautaki Kano Ārai Mate a te Kāwanatanga mō KOWHEORI-19, he kimi ara e puta ai i ngā here hauora tūmatanui, ā, kia mutu hoki te urutā
On 18 May 2020 – three months after New Zealand's first known COVID-19 case – Cabinet approved the COVID-19 Vaccine Strategy. The strategy aimed to obtain a 'sufficient quantity of a safe and effective vaccine' at an affordable cost.142
The former Prime Minister, Rt. Hon. Dame Jacinda Ardern, told us '… we always had a plan to transition away from alert levels into a reliance on the vaccine ...' and '… fundamentally vaccine is your exit.143 Former Finance Minister, Hon. Grant Robertson noted '… we had from the beginning of 2021 said that vaccination was our way out.144
Cabinet was advised that vaccination was a critical platform for reopening borders and leaving lockdowns, and could be the only way to bring the pandemic to an end.145 Officials advised Ministers that vaccines could reduce the severity of illness, prevent the pandemic overwhelming the health system, reduce transmission rates and so reduce the need for 'blunt tools like border controls and lockdowns'.146
Safety and effectiveness of vaccines underpinned the design and delivery of the Government's vaccine strategy and immunisation programme. Officials warned Cabinet that using an unsafe vaccine would 'undermine New Zealanders' willingness to be vaccinated for several years to come, and across a wider range of diseases than COVID-19.' They advised that only 'large well-designed studies that follow recipients of vaccines for a period of time' could provide assurance of safety and effectiveness. Medsafe would assess safety and efficacy and carry out ongoing active surveillance in accordance with its statutory role.147
We received submissions that the Government was wrong to rely so much on vaccination as a way out of the pandemic, and should have preferred alternative strategies to vaccination, such as relying on natural immunity, taking Vitamin D, or making healthy lifestyle changes.148 This view was almost always presented to us within the context of a belief that COVID-19 was not a serious illness with high mortality.
There is overwhelming international scientific evidence of significant rates of death and serious illness from COVID-19.149 Officials and scientists told us that relying largely on natural immunity against COVID-19 would have involved a significant burden of death and illness.150
We earlier outlined why vaccination is a highly successful public health strategy. The decision to adopt this as a strategy is not directly within our terms of reference. Nevertheless, we observe that the approach taken was appropriate, and consistent with overwhelming scientific advice.
The epidemiology of the COVID-19 virus is outside the scope of this inquiry.151 We have assessed the key decisions within our scope against the international scientific consensus that COVID-19 was a substantial threat to public health.
2.1.1.5 Purchasing vaccines | Te hoko kano ārai mate
The vaccine strategy was initially focused on securing vaccine supply and delivery. In August 2020, Cabinet decided to purchase a diversified portfolio of vaccines 'to increase the chances of securing safe and effective COVID-19 vaccines at the earliest possible time for New Zealand'.152 Target vaccines were prioritised based on expected performance, availability and access.153
The Government's eventual portfolio comprised four vaccine candidates from three leading technology platforms: Pfizer/BioNTech, AstraZeneca/University of Oxford, Janssen and Novavax.154 The Government announced its first Advanced Purchase Agreement in October 2020 with Pfizer, for the Comirnaty vaccine.155 Agreements with Novavax, Janssen and AstraZeneca followed in November and December 2020.156
2.1.2 What happened: pre-market approval of vaccines | Ngā mea i pā mai: te whakaaetanga o ngā kano ārai mate i mua i te hokohoko
Between February 2021 and October 2022, Medsafe took a series of decisions to approve vaccines to enable public access to a COVID-19 vaccine in New Zealand. These key decisions involved adapting the standard approval processes for the COVID-19 vaccines, giving provisional approval for the use of Comirnaty (first for adults, later for children aged 12–15 years and later still for those aged 5–11 years), and then giving provisional approval to other COVID-19 vaccines.157
| Table 1: Sequence and timing of approval process | |
|---|---|
| Date | Decision |
| Between October 2020 and February 2021 | Adaptation of approval processes |
| 3 February 2021 | Provisional approval of the Pfizer vaccine (Comirnaty) |
| 21 June 2021 | Approved use of Comirnaty extended to 12–15-year-olds |
| 7 July 2021 | Provisional approval of the Janssen vaccine (Jcovden)158 |
| 29 July 2021 | Provisional approval for the AstraZeneca vaccine (Vaxzevria) |
| 16 December 2021 | Provisional approval of the paediatric Comirnaty vaccine for 5–11-year-olds |
| 4 February 2022 | Provisional approval for the Novavax vaccine (Nuvaxovid) |
2.1.2.1 The legal framework for decisions on vaccine approval | Te tūāpapa ture mō te whakatau whakaaetanga mō ngā kano ārai mate
The Medicines Act 1981 requires the Minister of Health's consent or provisional consent before any medicine can be sold, distributed or advertised.159 This power is delegated to the Group Manager of Medsafe.160
A sponsor of a medicine (usually the pharmaceutical company) applies for either 'full' consent (under section 20) or 'provisional' consent (under section 23).161
Applicants for full consent must provide basic details such as the medicine's name, ingredients, dosage and use, as well as more substantive information derived from tests and reports about its safety and efficacy. Applicants also must provide evidence on whether the authorities in other countries had approved or consented to its distribution in the same form, and for the purpose, proposed for New Zealand.162
Applicants for provisional consent do not need to (but may) provide substantive information about safety and efficacy.163 Provisional consent is limited to two years or less, and Medsafe can impose conditions on it.164 The High Court has described the purpose of provisional consent as a way to authorise new medicines temporarily where there is a clear and immediate need for the medicine but a full consent process is not possible because 'all the information necessary to establish safety is not available'.165
Before giving approval, Medsafe must consider all the information provided with the application and other relevant matters.166 It must 'weigh the likely therapeutic value of the medicine against the risk (if any) of the use of the medicine injuriously affecting the health of any person.167 Medsafe may refer an application to an appropriate committee for advice, if it considers further advice and expertise is needed to make a decision (as happened for COVID-19 vaccines).168
2.1.2.2 Medsafe's approval processes are more comprehensive than legislation requires | He whānui ake ngā tukanga whakaaetanga a Medsafe i ngā whakaritenga a te ture
In its standard process for approving new medicines (which in most aspects is more comprehensive than legislation strictly requires), Medsafe first requires applicants to provide administrative and prescribing information, like the name of the product, dose and labelling information, a datasheet and package insert for consumers.169 Medsafe also requires information on manufacturing and storage processes and on the chemical and biological make-up of the medicine.
Applicants must, in addition, provide data on non-clinical studies, relevant literature studies, and from clinical trials, as well as on overseas approvals and on how the safety of the medicine will be monitored. Medsafe evaluates the quality and comprehensiveness of the clinical data, taking account of the type of medicine, but does not require a set number of clinical trials.170
Medsafe staff are experts in pharmacy, medicine and related sciences. They evaluate the application against criteria that draw on international best practice.171 After several layers of peer review, their reports go to Medsafe's Group Manager for a decision. If Medsafe considers the information to be inadequate, it requests further information from the sponsor.
As required by the Medicines Act, for both full and provisional approval, Medsafe assesses whether the safety profile of the medicine is acceptable and the benefits outweigh the risks.
For benefits it considers:
- has efficacy been demonstrated in the target population of people who will use the medicine?
- is the medicine significantly better than a placebo?
- how does the medicine compare to any alternative treatments?
- how many people have the condition that this medicine will treat or prevent?
- what is the natural history of the disease?
- is [the disease] self-limiting, chronic or fatal?
For risks it considers:
- what proportion of people taking the medicine experience an adverse reaction?
- how many of these adverse reactions are considered to be serious?
- how many people stopped treatment because of an adverse reaction?
- are the adverse reactions reversible, treatable or avoidable (for example, interactions with other medicines)?172
Medsafe bases its assessment of an application on the information provided and does not independently test medicines. However, it required independent testing of each batch of COVID-19 vaccine supplied in New Zealand.
Medsafe gave provisional consent to 33 medicines in the five years before COVID-19, usually to enable the ongoing supply of critical funded medicines to support a national stock shortage, but sometimes to enable Medsafe to place conditions on approval or to meet an otherwise unmet clinical need.173 An example of when Medsafe granted provisional consent to a vaccine is the novel MeNZB vaccine in July 2004, when there was a high clinical need to manage the outbreak of meningococcal disease, and provisional consent was granted even though the vaccine had not completed Phase III clinical trials.174
2.1.2.3 Medsafe made a suite of changes to its approval process for COVID-19 vaccines | I whakarerekē a Medsafe i ētahi wāhanga o te tukanga whakaaetanga mō ngā kano ārai mate KOWHEORI-19
In 2020, Medsafe developed plans to evaluate any vaccine as quickly as possible without compromising a robust evaluation.175 From September 2020, it participated in a series of strategic meetings with members of the International Coalition of Medicines Regulatory Authorities to align international COVID-19 policies and speed up the development, approval and availability of COVID-19 vaccines.176
Alongside the vaccine taskforce led by the Ministry for Business, Innovation and Employment, Medsafe began early discussions with pharmaceutical companies to understand their timeframes, technologies and deployment intentions. This helped Medsafe to prioritise applications, and to plan for teams to quickly assess vaccine candidates.177
Because of the significant clinical need for safe and effective vaccines against COVID 19, Medsafe gave priority to all applications for COVID-19 vaccines and streamlined its administrative process for that purpose.178 Its Group Manager was more closely involved in the process than usual.179 Medsafe assessed applications in the order in which they were received from sponsors, allowed rolling applications so companies could submit different sections of the required dossier of information as the data became available, and evaluated each section as it was received.180 This meant it could make a final recommendation much sooner than usual, while ensuring all the usual steps had been completed.181
Because of the novelty of some of the vaccines' technology and high public interest, the Group Manager established a COVID-19 Vaccine Advisory Group on 4 November 2020 to support the approval and assessment process.182
2.1.2.4 Medsafe granted provisional approval of Comirnaty in February 2021 | I whakawhiwhi a Medsafe i te whakaaetanga rangitahi mō te Comirnaty i Pēpuere 2021
After discussions beginning a month earlier, Pfizer applied to Medsafe for provisional consent for its Comirnaty vaccine in October 2020. In November 2020, on Medsafe's request, Pfizer revised its application to be for full consent, which meant that substantive evidence on safety and efficacy was required.
Because Comirnaty uses mRNA, on 29 January 2021, Pfizer asked the Environmental Protection Authority, which regulates genetically modified organisms in New Zealand, to determine whether the Comirnaty vaccine was a new 'organism' for the purpose of the Hazardous Substances and New Organisms Act 1996. The Authority concluded that it did not need to approve the use of Comirnaty as it did not meet the definitions of 'organism' under that Act. This was because the mRNA molecule in Comirnaty contains only the genetic information to enable the production of the spike protein – Comirnaty does not contain any genes allowing it to replicate itself.183 Appendix 1 discusses this in more detail.
After a 'vigorous assessment' of Pfizer's application between October 2020 and January 2021, Medsafe granted provisional consent for Comirnaty, on 3 February 2021.184 The consent was for nine months and with 58 conditions requiring Pfizer to produce more data on quality and safety within specific timeframes.185 Approval was for use in people aged 16 and over, as the vaccine's safety and efficacy had not yet been established for younger age groups (but was being tested as part of the ongoing clinical trial).186
Pfizer provided a dossier to support its application that included non-clinical studies (in vitro and in vivo), data from a combined Phase I/II study and a combined Phase II/III study (both of which were ongoing), and confirmation that regulators in the European Union and Canada had approved Comirnaty with conditions.187
We have seen from the documents that the clinical data reassured Medsafe, by showing that two doses of the Comirnaty vaccine administered three weeks apart provided 95 percent protection against symptomatic COVID-19.188 Increasing international experience provided greater assurance of Comirnaty's safety. Most reactions to the vaccine were mild or moderate in intensity and resolved within a few days after vaccination.
Severe adverse events were reported in 1.2 percent of the trial group, which is comparable to other licenced vaccines.189 Importantly, adverse events in clinical trials can be coincidental or due to the 'nocebo effect' (a phenomenon where a negative expectation about a treatment or substance can lead to negative side effects or a worsening of symptoms, even if the treatment is inert or ineffective). Of the participants in the Pfizer trials who were given the placebo, 0.6 percent experienced severe adverse events, so the actual rate of severe adverse events that may have been related to the administration of Comirnaty during the trial is 0.6 percent.190
Even so, Medsafe did not recommend granting full consent for Comirnaty because some quality and clinical issues remained unresolved.191 Due to the speed of development and the expected modifications for large scale manufacture, substantially less quality data existed than in a standard application. Data on efficacy was also lacking, including on the duration of protection (data was then only available for up to two months after vaccination), and in subpopulations including Māori, Pacific and Asian peoples, immunocompromised people, pregnant and lactating women, and people who had had COVID-19. Data was lacking on protection against severe disease, prevention of asymptomatic infection and disease transmission, and new variants. Medsafe noted that data on long-term safety (including rare and delayed adverse events) was not available (this was not unusual for a new medicine, as discussed in section 2.1.3.2).
Although Medsafe did not recommend full consent, it recommended seeking independent advice on provisional approval, based on high clinical need, rapidly accumulating international experience, and an expectation of further data from Pfizer around April 2021. They noted the effectiveness of vaccines as a public health strategy and observed that public health measures other than vaccines can be socially disruptive and have adverse economic consequences.192
2.1.2.5 Medsafe consulted advisory bodies and regulators in New Zealand and overseas before giving approval | I rapu kōrero a Medsafe i ngā rōpū tohutohu me ngā kaiwhakahaere ture i Aotearoa me tāwāhi i mua i te tuku whakaaetanga
While Medsafe was reviewing Pfizer's application, they sought advice from experts, including their international partners in Australia, Canada and the European Union.
In January 2021, Medsafe sought advice from the COVID-19 Vaccine Advisory Group on Pfizer's application, including about the risks of only having two months' safety and efficacy data.193 The Group said that two months of follow-up data was not ideal, but more data could be provided after the medicines were approved. Although two months' data might not reveal risks to specific population cohorts or longer-term safety effects, it was enough to identify severe adverse events, as most adverse events considered plausibly linked to vaccination generally start within two months of receiving a vaccine. The Group considered two months' efficacy data was sufficient to determine the initial protective period, and short-term protection was useful to control outbreaks and protect high exposure groups. Any risk posed by a lack of data about transmission or waning immunity could be mitigated by booster doses. They recommended careful ongoing monitoring of experience with the vaccine.
In the same month both the COVID-19 Vaccine Advisory Group and the Medicines Adverse Reactions Committee assessed Pfizer's Risk Management Plan.194 The Committee recommended asking Pfizer to amend specific provisions about collecting information on the use of the vaccine in patients with specific conditions and use of the vaccine with other common vaccines. Medsafe accepted this recommendation.195
The Ministry of Health's Science and Technical Advisory Group advised Medsafe on the transmissibility of a new variant of COVID-19 in the United Kingdom – relevant to its assessment of Comirnaty's risks and benefits.196 The Group advised that, as the new variant was more transmissible, rapidly deploying a vaccine could help protect vulnerable groups.197
Medsafe's Australian equivalent, the Therapeutic Goods Administration, shared its evaluation reports of Pfizer's application to use Comirnaty in Australia.198 The Administration gave Comirnaty provisional registration in January 2021.
Following Medsafe's recommendation that a provisional consent might be appropriate, in February 2021 it asked the Medicines Assessment Advisory Committee to review the application for consent, considering the data limitations.199 The Committee unanimously agreed with Medsafe's proposal to grant provisional consent to use Comirnaty for a period of nine months.200 This time limit would ensure that Pfizer met all its deadlines to supply data after receiving provisional consent.201 It recommended amendments to Medsafe's proposed conditions, and that the Comirnaty datasheet include information on provisional consent and its implications.202
The Committee also noted that risks to subpopulations would be managed as part of the Ministry of Health's immunisation programme and would require careful safety monitoring after use of the vaccine began.203 It considered, based on clinical trials, that it was 'not unreasonable to assume there is no genetic reason for different responses in different ethnic groups.204
The Medsafe Group Manager accepted this advice and granted provisional consent on the basis that the therapeutic value of the vaccine outweighed potential health risks for people using it. In reaching that decision, the Group Manager was satisfied that the vaccine could offer important protection against COVID-19, it was adequately safe and effective and that giving provisional consent would offer time limits and conditions to enable Medsafe to monitor the vaccines and make changes as necessary. The Medicines Assessment Advisory Committee's support of the provisional consent gave Medsafe confidence to make the decision.205
Medsafe communicated its decision to the public during a live media stand-up that was also livestreamed on 3 February 2021, accompanied by a written media statement and updates to its website.206
2.1.2.6 Medsafe extended approval to children and renewed provisional consent for Comirnaty | I whakawhānui a Medsafe i te whakaaetanga ki ngā tamariki, ā, i whakahou anō i te whakaaetanga rangitahi mō Comirnaty
Medsafe approved use of Comirnaty for children aged 12 to 15 years on 21 June 2021.207 The approval retained the conditions of the original consent that had not yet been met, with some amended timeframes.
Pfizer had provided further clinical data on Comirnaty's safety and efficacy for this age group. This showed that the vaccine was generally well tolerated by adolescents and that the rate of common reactions was like that for adults. Medsafe noted that COVID-19 was causing morbidity and mortality among adolescents and that the vaccine provided a high level of protection.208
On 28 October 2021, Medsafe renewed the provisional consent for Comirnaty for another two years, from 3 November 2021.209 The conditions on the initial provisional consent had been fulfilled by this time, but some conditions were retained that required the ongoing supply of clinical trial data, batch testing and the monitoring of the vaccine.210 On 8 November, Medsafe amended the approval of Comirnaty to also include a booster dose.211
On 16 December 2021, Medsafe's Group Manager gave provisional consent to the paediatric Comirnaty vaccine – a different formulation and smaller dose than the adult vaccine.212 The vaccine was for children aged five to 11 years with the expiry date for the provisional consent aligning with that of the renewed consent for the adult vaccine.
Medsafe had reviewed Pfizer's information and data on the paediatric vaccine and concluded that the vaccine's quality and safety was comparable to the adult vaccine. The data showed that it was generally well-tolerated by its intended age group and was 91 percent effective in reducing symptomatic infection in clinical trials, following two doses.213
Medsafe sought the Medicines Assessment Advisory Committee's advice on Pfizer's application because of some data limitations and the public interest.214 On 14 December 2021, the Committee unanimously recommended provisional consent for the paediatric vaccination.215 Medsafe also sought and received agreement from the Medicines Adverse Reactions Committee to consider Medsafe's questions to Pfizer on its Risk Management Plan for Comirnaty and suggest any changes.216
In weighing the vaccine's risks and benefits, the Medsafe Group Manager assessed that COVID-19 was a highly contagious disease that could lead to hospitalisation and death in children, so there was a clinical need for immunisation in this age group.217 Medsafe set conditions like those for the adult vaccine to require more information on quality, safety and efficacy as it became available.218
2.1.2.7 Medsafe gave provisional approval to other COVID-19 vaccines in 2021 and 2022 | I whakawhiwhi a Medsafe i te whakaaetanga rangitahi ki ētahi atu kano ārai mate KOWHEORI-19 i ngā tau 2021 me 2022
On 8 December 2020, Medsafe received an application from Janssen for their COVID-19 vaccines. This was followed by an application on 29 January 2021 from AstraZeneca and one on 19 February 2021 from Novavax.
The process of approval for the AstraZeneca, Janssen and Novavax vaccines followed the same general process as for the Comirnaty vaccines, including prioritising the applications over other medicines, assessing the applications on a rolling basis as data became available, and requesting review of the application by the Medicines Assessment Advisory Committee.219
From March 2021, and before Medsafe's approval of those vaccines, international reports emerged of links to thromboembolism, Guillain-Barré Syndrome and capillary leak syndrome rarely associated with the adenovirus vaccines (AstraZeneca and Janssen).220 Most other side effects were mild. Medsafe's team highlighted those safety concerns to the Group Manager, and the Medicines Assessment Advisory Committee recommended that the use of those vaccines should be restricted to circumstances where no other approved COVID-19 vaccine was available or clinically indicated.221
With each of the three vaccines, Medsafe applied the statutory test that the benefits must outweigh the risks. Where information was not yet available, including in relation to the upscaling of production, it considered that the additional information could be requested within specific timeframes as conditions on a provisional consent. It also proposed conditions requiring ongoing safety monitoring and reporting, and it took into account the decisions of international regulators to approve the vaccines. The Medicines Assessment Advisory Committee supported each of the proposals to give provisional, time-limited consent with conditions.222
The Medsafe Group Manager gave provisional consent to the Janssen vaccine on 7 July 2021.223 This was followed by provisional consent to the AstraZeneca vaccine on 29 July 2021224 and the Novavax vaccine on 4 February 2022.225 Each provisional consent was for nine months, with conditions – including a condition that the vaccine could only be sold to the New Zealand Government and distributed in accordance with the Government's COVID-19 vaccine rollout or donated to other countries. Datasheets for the Vaxzevria and Janssen vaccines highlighted the risks of vaccine-induced thrombotic thrombocytopenia or thrombosis with thrombocytopenia syndrome, capillary leak syndrome and Guillain-Barré syndrome.226
Following legal challenge, the legislation for provisional approvals was amended
In April 2021, Ngā Kaitiaki Tuku Iho Medical Society Inc (Ngā Kaitiaki) sought a judicial review to challenge Medsafe's decision to grant provisional consent to Comirnaty.227 The challenge was on the basis that the Medicines Act only permitted provisional consent if the medicine was to be used on a restricted basis for the treatment of a limited number of patients.228
Ngā Kaitiaki argued that Medsafe's provisional approval was 'an error of law, as it did not identify criteria for identifying the "limited number of patients" the provisional consent applied to as required by section 23(1).' In its judgment delivered on 18 May 2021, the High Court agreed, as the Government intended to make Comirnaty available to all New Zealanders over 16 years of age. However, the Court declined to grant interim orders due to the adverse public and private repercussions. The Court also considered that the process for provisional approval was robust and responsive in practice, and that this meant that any risks associated with provisional consent were 'considerably diminished'.229
Anticipating the judgment in the Ngā Kaitiaki case, Cabinet agreed on 17 May 2021 to amend the Medicines Act 1981 to validate the provisional consent of the Comirnaty vaccine; and 'to clarify the purpose and scope of provisional consent consistent with modern international regulatory practice for medicines.230
Parliament amended the Medicines Act 1981 on 25 May 2021.231 The new section 23(1) permits the Minister to give provisional consent 'to the sale or supply or use of a new medicine if the Minister is of the opinion that it is desirable that the medicine be sold, supplied or used' – removing the words 'on a restricted basis for the treatment of a limited number of patients'. The Amendment Act retrospectively validated the provisional consent for Comirnaty.232
The applicants in MKD v Minister of Health challenged the decision to grant provisional approval for the paediatric vaccine.233 They argued it should be reconsidered in the light of expert evidence demonstrating that the risks of the paediatric vaccine outweighed the benefits. The challenge was unsuccessful as the Court considered Medsafe's Group Manager had properly applied the statutory test and made an informed decision that was reasonably open to him.234
2.1.3 Key decisions: pre-market approval of vaccines – our assessment | Ngā whakatau matua: te whakaaetanga o ngā kano ārai mate i mua i te hokohoko – tā mātou aromatawai
2.1.3.1 Medsafe adapted its processes to be agile and responsive | I urutau a Medsafe i ngā tukanga kia kakama, kia urupare hoki
Medsafe was agile and adaptive in its regulation of COVID-19 vaccines during the pandemic. This allowed rapid approval of a vaccine without taking short cuts. The High Court considered that the process for approving the vaccines went 'above and beyond' the usual provisional consent process, and that 'it is difficult to see how the assessment process could, in the circumstances, have been more thorough.235 We endorse those comments.
Medsafe balanced the right factors in making decisions, informed by experience in comparable jurisdictions, and especially through collaboration with the International Coalition of Medicines Regulatory Authorities and the Therapeutic Goods Administration in Australia. The pace of Medsafe's decision-making was broadly in step with other international regulators. Medsafe assessed Pfizer's application between October 2020 and early February 2021, a timeframe comparable to Australia's evaluation, between October and January, and to that of Canada, the European Union and the United Kingdom, which completed their reviews between October and December 2020.236 The decisions to prioritise COVID-19 vaccines showed appropriate consideration and balancing of the effects on health and wider society if a vaccine was not approved as quickly as possible in response to the identified clinical need.
2.1.3.2 Medsafe's provisional approval of Comirnaty was sufficiently well-informed | He tino whai mōhio te whakaaetanga rangitahi a Medsafe mō Comirnaty
We consider that Medsafe was sufficiently well-informed in its decision to grant provisional approval of Comirnaty. The legislative test required Medsafe to be satisfied that the benefits of Comirnaty outweighed its risk, acknowledging that all medicines carry some risk.237 Medsafe had sufficient information to confirm the vaccines' safety and efficacy against COVID-19, based on the interim results of ongoing clinical trials and the experience of the rollout overseas. Its provisional approval of Comirnaty reflected the advice it had received from its staff, from comparable jurisdictions and from domestic advisory bodies. The advice provided enough information to make judgements about the risks and benefits of the vaccine, in the context of the potential health, social and economic disruption of a pandemic (and response) without a vaccine.
We acknowledge that there were obvious data limitations – more than would usually be expected due to the speed of the vaccine's development. This has been a source of significant concern for some submitters. We consider that Medsafe's decision to approve Comirnaty, despite those data limitations, was sufficiently informed.
Data gaps (particularly in long-term safety and efficacy data) are expected for new medicines on approval. Clinical trials are tested on groups of people that are not always reflective of all the world's different populations. That is why mechanisms exist for monitoring medicines safety after they enter the market. Clinical trials continue to produce data for review at several points after approval (discussed further in Appendix 1). Medsafe's management of vaccine safety did not end with approval but was an ongoing process, subject to continual review and reassessment (see section 2.1.6). These efforts were supported by the rapid availability of international data, as populations around the world were immunised.
Giving provisional rather than full approval of Comirnaty provided the opportunity and framework to obtain further information on safety and efficacy as experience with the vaccine grew. Medsafe was reassured that the next tranche of safety and efficacy trial data was due three months after approval, in April 2021, and manufacturing data was required within specific timeframes. Medsafe put measures in place through conditions, review of risk management plans and pharmacovigilance, to mitigate risk, and had the power to revoke vaccine approval if necessary. As a result, Medsafe was in a position to act quickly if new information emerged.
Medsafe's decision-making framework is designed to be flexible and takes clinical need into account in its risk-benefit analysis when deciding on the degree and type of information needed to support approval of a medicine. This framework is entirely appropriate given future pandemics will have their own unique characteristics.
We acknowledge that some people disagree with Medsafe's decision that the benefits of Comirnaty outweighed its risks. They consider the risks of the vaccine then, and now, well outweigh any perceived benefit. We do not agree with that view and discuss some of those perceived risks in more detail in Appendix 1.
This belief is usually underpinned by a perception that COVID-19 did not present a significant threat to public health. As noted above, the evidence does not support this perception. In any event, our task is to assess whether Medsafe's decision was sufficiently informed, based on the process it used and the information available to it. Having reviewed Medsafe's key decision to approve Comirnaty, we share the Court's view that it is difficult to see how the process and information leading to the decision could have been more thorough.
2.1.3.3 The approvals of the other COVID-19 vaccines were sufficiently well-informed and did not produce unforeseen consequences | I hāngai pai te mōhiohio mō ngā whakaaetanga o ētahi atu kano ārai mate KOWHEORI-19, ā, kāore i hua ake he pānga ohorere
Medsafe's decisions to grant provisional consent to the AstraZeneca, Janssen and Novavax vaccines were also sufficiently well-informed. Medsafe undertook a careful assessment of the available information and data, including from international regulators, sought additional advice from the Medicines Assessment Advisory Committee, and its decisions reflected advice. Despite the risks presented by the data limitations and potential adverse reactions, the vaccines were effective against COVID-19 and their approval gave the public options for different COVID-19 vaccines. The risks were carefully mitigated by time-limited provisional consent with conditions, ongoing pharmacovigilance and clear information published in the publicly available datasheets.
We are satisfied that it was open to Medsafe to conclude on the information available to it that the benefits of the AstraZeneca, Janssen and Novavax vaccines outweighed their risks, and to grant provisional approval. We are not aware of any resulting unforeseen consequences.238
2.1.4 Agreeing to use vaccines in the immunisation programme | Te whakaae ki te whakamahi i ngā kano ārai mate i roto i te hōtaka ārai mate
Once Medsafe approved COVID-19 vaccines, Cabinet was responsible for policy decisions to use them in the Government's immunisation programme. The Government considered that a widely-vaccinated population was the COVID-19 pandemic 'exit plan'.239 Cabinet made decisions to impose and lift restrictions, such as lockdowns and mandates, contingent on most people being vaccinated. Decisions on how vaccines were rolled out are therefore important context for assessing several key decisions covered by our Terms of Reference.
However, as Phase One of the Inquiry reviewed the vaccine rollout as the primary focus of its chapter on vaccination, our review here is narrow.240 Given that the rollout took 10 months to achieve 90 percent coverage of the eligible population, Phase One concluded that New Zealand's rollout was 'very effective' and achieved greater and quicker coverage than many comparable countries.241 Phase One referred to reviews by the Office of the Auditor-General and the Waitangi Tribunal of the vaccine rollout.242
The key findings stemming from those reviews relate to missed opportunities to ensure the vaccine reached everyone (particularly Māori and Pacific peoples) equally quickly. As a result, when the Delta outbreak of COVID-19 emerged in August 2021, rates of vaccination for Māori and Pacific peoples were substantially lower than for other population groups. Phase One found that if this had been effectively addressed earlier, New Zealand may have reached 'its immunisation target earlier, allowing lockdowns and other stringent restrictions to be relaxed sooner.243 The Auditor-General made a number of findings early on in the rollout, which were subsequently addressed by the Ministry of Health before the wider rollout began. The Auditor-General noted the need for effective communication about the vaccine roll-out with stakeholders and the public, to build confidence and manage expectations.
We do not duplicate the work of Phase One and other reviews. We endorse their findings. We have limited our review of how the vaccines were rolled out to two key decisions: the decision to approve the Comirnaty vaccine as the primary vaccine, and the decision to approve the use of other vaccines as second-line options for people who were unable or unwilling to receive the Comirnaty vaccine and to manage supply risks.244
2.1.4.1 Cabinet agreed to use Comirnaty as the first-line vaccine | I whakaae te Rūnanga Minita kia whakamahia a Comirnaty hei kano ārai mate tuatahi
On 9 February 2021 Cabinet agreed to the use of Comirnaty in the immunisation programme for those aged 16 and over, starting with border and managed isolation and quarantine workers and their household contacts, and then expanding to wider groups as supply allowed from March.245
Cabinet received advice from the Ministry of Health, the COVID-19 Vaccine Technical Advisory Group and Health Ministers before making its decision.246 Advice highlighted the data limitations and clinical conditions Medsafe had attached to the vaccine and provided additional recommendations about how it should be used. It noted that Comirnaty was suitable for use in all people aged 16 years and over, including those who were immunocompromised, pregnant, or breastfeeding, except those who had experienced a previous allergic reaction to a component of the vaccine.247 It recommended observing all recipients for 30 minutes after being vaccinated, and that they receive information about expected responses and how to manage them.
Cabinet accepted the advice but left room to evaluate the use of Comirnaty if future vaccines were going to be more suitable or effective.248 It later agreed to use Comirnaty in children aged 12 to 15 years in August 2021249 (see section 2.1.7.6) and in children aged 5–11 years in December 2021.250 This followed further regulatory approval and advice from the Ministry of Health, the COVID-19 Vaccine Technical Advisory Group and a child wellbeing impact assessment.251 Cabinet followed advice from the COVID-19 Vaccine Technical Advisory Group and the Ministry of Health that mandates, vaccine certificates or vaccine targets should not be used for the 5–11 year old age group, and children should not be denied access to locations or events based on their vaccination status.252
2.1.4.2 Cabinet made a series of decisions to use second-line vaccines in the immunisation programme | I whakatau te Rūnanga Minita i tētahi raupapa kaupapa kia whakamahia ngā kano ārai mate tuarua i roto i te hōtaka ārai mate
Cabinet agreed to use other COVID-19 vaccines as second-line options in late 2021 and early 2022.253 Cabinet did so to better manage supply risks, to provide greater resilience to new variants of the COVID-19 virus and to provide people with non-mRNA alternatives to Comirnaty, with the goal of increasing vaccine uptake among those reluctant to receive such vaccines.254
The first second-line vaccine to be approved for use was the Janssen vaccine on 22 September 2021. However, supply issues prevented its timely arrival in New Zealand. This influenced Cabinet's decision on 8 November 2021 to use the AstraZeneca Vaxzevria vaccine,255 which became available on 26 November 2021.256 Cabinet agreed to use the Novavax vaccine on 28 February 2022 – this was two months after the 90 percent vaccination target had been achieved.
In each case, Cabinet considered technical advice from the COVID-19 Vaccine Technical Advisory Group like that provided for decisions on the Comirnaty vaccines.257 Also, in each case, the Group advised there was no indication for the widespread use of the other vaccines, as the Comirnaty vaccine was meeting the needs of the immunisation programme well. Cabinet was advised that making another vaccine available could contribute to the success of the immunisation programme in reaching a target of vaccinating 90 percent of the eligible population.258
Cabinet retained Comirnaty as the preferred vaccine, as none of the others offered a materially improved level of safety, efficacy or supply.259 Cabinet also recognised that introducing new vaccines into the immunisation programme would present challenges for logistics, training, access and communication.260
By 28 November 2022, more than 12 million doses of Comirnaty had been administered. Due to their later availability, the uptake of the second-line vaccines was comparatively very low – about 9,000 doses of AstraZeneca were administered, and 7,500 doses of Novavax. We saw no evidence of the Janssen vaccine being administered.261
2.1.5 Key decisions to use vaccines in the immunisation programme – our assessment | Ngā whakatau matua mō te whakamahi i ngā kano ārai mate i te hōtaka ārai mate – tā mātou aromatawai
2.1.5.1 Comirnaty was a good choice for a first-line option | He kōwhiringa pai te Comirnaty hei kōwhiringa tuatahi
Three key factors governed Cabinet's decisions to use vaccines in the Immunisation Programme: regulatory approval, supply and safety. Cabinet chose Comirnaty as the first-line option because Medsafe approved it first, it was sufficiently safe and effective and Pfizer was able to supply it to the New Zealand market early on and scale-up volume over time.
The decision-making process was appropriately agile. It left room for flexibility if there were supply disruptions, unforeseen side-effects from the vaccines, or different responsiveness to emerging strains of the virus.
The use of Comirnaty as the first-line option was broadly successful in achieving high levels of vaccination in the population and so protecting New Zealanders from the worst impacts of COVID-19 (Phase 1 Report 7.3.1). We were told that New Zealand was fortunate in choosing Comirnaty as its first and primary option. Comirnaty was effective and met safety standards. Other vaccines either were not materially better than Comirnaty in these respects or were less effective.262
2.1.5.2 Second-line vaccines would ideally have been made available before mandates were imposed | He mea pai ake, mena kua wātea kē ngā werohanga rārangi-tuarua i mua i te whakatakoto o ngā whakature
Second-line vaccines provided options for those who were unable or unwilling to receive the Comirnaty vaccine. However, Vaxzevria, the first available second-line vaccine, only arrived in New Zealand in late November 2021, after the Government had put in place several vaccine mandates.263 As a result, people subject to mandates did not have a choice of vaccine until late 2021.
New Zealand was pursuing many different COVID-19 vaccines that were under development, and the Government had the foresight to purchase a portfolio of different vaccines based on different technologies. In future pandemics, there might only be one vaccine available. However, if possible, we consider that offering individuals alternatives to choose from within a government immunisation programme will support the ability to implement vaccine mandates with greater public acceptance.
2.1.6 What happened – post-approval vaccine safety | Ngā mea i pā – te haumaru o ngā kano ārai mate i muri i te whakaaetanga
As described, when new medicines are introduced, evidence about safety and efficacy is always based on data from trials, rather than population use. Rare adverse events therefore do not usually appear until a medicine is used on a population-wide basis. Accordingly, a medicine's safety continues to be monitored and assessed after it is distributed.
The Government considered that ensuring the safety of COVID-19 vaccines was critical to the success of the immunisation programme.264 In this section, we assess how the Ministry of Health made or led a key set of decisions about how it would monitor and report on the safety of COVID-19 vaccines once they began to be rolled out. We illustrate how this worked in practice with a description of how Medsafe began an investigation in April 2021 into international reports of myocarditis as a very rare side effect to mRNA COVID-19 vaccines, and how from April 2021 health authorities began acting on identified concerns.265
We begin by describing the international, legal and administrative framework for monitoring and acting on concerns about vaccine safety in New Zealand, after a vaccine has been approved and begun to be used.
2.1.6.1 Monitoring vaccine safety is an international collaborative effort | He mahi ngātahi ā-ao te aroturuki i te haumaru o ngā kano ārai mate
New Zealand is a member of the World Health Organization Programme for International Drug Monitoring which collects, appraises and shares information on serious reactions to vaccines and other drugs.266 The Programme works in close collaboration with the Uppsala Monitoring Centre, which maintains a global database of adverse event reports (VigiBase).267 Member states, including New Zealand, contribute individual case safety reports. The independent Global Advisory Committee on Vaccine Safety oversees evidence on vaccine safety performance and advises the World Health Organization on safety issues.268
At the time COVID-19 vaccines were being rolled out, the international approach to pharmacovigilance largely relied on spontaneous reporting of adverse events. In describing what such systems should do, the World Health Organization noted the following functions:269
- to collect reports of adverse drug reactions
- to identify safety signals. This is particularly important for newer drugs where potential adverse events may be unknown or poorly characterised
- to assess the risk associated with specific drugs and options for managing that risk, for example, communication of potential adverse reactions, review of drug use
- to provide effective communication on the safety of specific drugs 'including dispelling unfounded rumours of toxicity attributed to medicines and/or vaccines'.
2.1.6.2 The Medicines Act sets out requirements for pharmacovigilance | E tautuhi ana te Ture Rongoā i ngā whakaritenga mō te aroturuki haumaru o ngā rongoā
In New Zealand, Medsafe has overall responsibility for pharmacovigilance with the Group Manager holding delegated powers under the Medicines Act 1981.270
Medsafe can require proactive safety monitoring by placing a condition on the consent.271 This is what happened for the COVID-19 vaccines, where Medsafe required the sponsors to perform pharmacovigilance activities and provide periodic safety reports. The legal framework permits or requires action from either Medsafe or the sponsor once they become aware of issues.
The actions available to the government under the Medicines Act if there are safety concerns include requiring sponsors to provide further information or assurances about safety, prohibiting or placing conditions on the sale or supply of the medicine, or revoking or suspending consent to a medicine.272
2.1.6.3 Medsafe oversees a comprehensive system for receiving and assessing reports of adverse events | Ka whakahaere a Medsafe i tētahi pūnaha whānui mō te whiwhi me te aromatawai i ngā pūrongo mō ngā pānga kino
Figure 1: Adverse event reporting and safety signal assessment
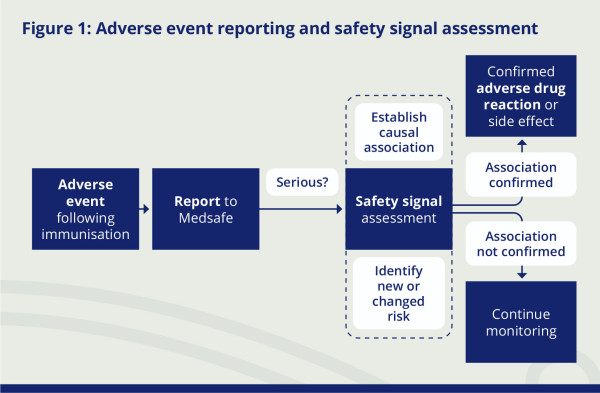
In New Zealand, the Government monitors the safety of medicines primarily through assessing reports of adverse events, as is the usual practice internationally (see section 2.1.6.1).273
The Ministry of Health contracts the Centre for Adverse Reactions Monitoring to collect and evaluate spontaneous reports of suspected adverse events to medicines. The Centre's programme is a voluntary system and any person can make a report.274 Both before and after the pandemic, New Zealand had one of the highest rates in the world of reporting per capita.275
A report of an adverse event is not a safety signal in and of itself. This is because not all adverse events will be caused by the medicine, so the assessment of safety signals considers if there is a causal relationship between the medicine and the reported adverse event.276 As explained below, in their process for managing safety signals, the Centre and Medsafe seek to determine if there are new risks associated with a medicine, or if the known risks have changed.277
On receipt of a report, the Centre's medical assessors prioritise more serious adverse events.278 An adverse event following immunisation is classified as 'serious' if it:279
- is a medically significant event or reaction
- requires hospitalisation or prolongs an existing hospitalisation
- causes persistent or significant disability or incapacity
- is life-threatening
- causes a congenital anomaly or birth defect, or
- results in death.
The assessors use medical and scientific judgment to identify 'medically significant' events.280 During the COVID-19 pandemic, they compiled a list of 'critical terms' that they considered could be significant and should trigger a report to an assessor for review.281
For serious reports, the Centre notifies Medsafe of any issues of concern, after evaluating the likelihood of an association between the medicine and the adverse reaction using causality assessment criteria standardised by the World Health Organization.282 In assessing causality, the Centre and Medsafe consider the time between receipt of the medicine and an event and compare the number of reports for an event with the expected rate in the relevant subpopulation, adjusting for seasonality.283
Adverse reactions related to vaccinations require careful assessment, especially if the vaccinations target a large proportion of the population or a selected group, such as older people or people who have pre-existing conditions. In both cases the likelihood of an adverse reaction being coincidental is increased, and it is important to distinguish coincidental events from real adverse reactions to the vaccines. For example, among 10 million vaccinated individuals, it can be predicted that 5.75 cases of sudden death (death within one hour of symptom onset) and 21.5 cases of Guillain-Barré syndrome would coincidentally occur within six weeks of vaccination.284
After its assessment, Medsafe may take no action, or one or more of: continued monitoring; a request for additional information from the sponsor; requesting an update of the datasheet; an instruction to sponsors to communicate directly with healthcare providers; seeking advice from the Medicines Adverse Reactions Committee; publishing a safety communication; suspending the distribution of medicine while investigations continue; or revoking consent for the medicine to be distributed.285 A safety communication may be either a monitoring communication for new identified safety concerns under review, or an alert communication if the review of the medicine identifies a causal link between an adverse event and the medicine.286
Medsafe also identifies potential safety issues received from other sources. These include reports from overseas regulators, New Zealand case reports, and the medical literature. Pharmaceutical companies and sponsors of medicines more generally must have their own pharmacovigilance system and may need to submit a Risk Management Plan, as was the case for COVID-19 vaccines.287
2.1.6.4 The Ministry of Health developed a strategy to strengthen pharmacovigilance for the COVID-19 vaccines | I whakawhanake te Manatū Hauora i tētahi rautaki ā-motu hei whakapakari i ngā tukanga haumanu-tiaki mō te aroturuki haumaru o ngā kano ārai KOWHEORI-19
In May 2020, as part of its COVID-19 Vaccine Strategy, the Government identified a need to strengthen the existing system to monitor vaccine safety. Rapid development of the vaccines and the use of new mRNA technologies highlighted the need for vigilance.288
In November 2020, Medsafe, on advice from the New Zealand Pharmacovigilance Centre, recommended a plan to strengthen existing arrangements to monitor COVID-19 vaccine safety and so maintain public trust and confidence in the immunisation programme.289 Medsafe's advice built on New Zealand's successful experience with surveillance of the safety of the novel MeNZB meningococcal vaccine between 2004 and 2007.290
Medsafe recommended continuing to use existing systems, making improvements to those systems, and introducing new tools, to ensure a strengthened system was in place. It highlighted effective communication and transparency as a key component.291 The plan evolved as it was implemented and the eventual system is outlined below.292
2.1.6.5 Pharmaceutical companies were required to proactively monitor safety | I herea ngā kamupene rongoā kia aroturuki haumaru i runga i te mātāra me te kaha
As outlined in Part 2.1.2.4, when Medsafe gave provisional consent to the COVID-19 vaccines, conditions were imposed to ensure the pharmaceutical companies proactively monitored safety. Medsafe required the sponsors to perform pharmacovigilance activities and interventions, produce periodic safety reports, submit Risk Management Plans, and produce documentation for health professionals and consumers about safe use of the vaccines and their risks, which was published on the Medsafe website.293
2.1.6.6 Specialist advisory groups supported the monitoring and assessment of the safety and effectiveness of COVID-19 vaccines | I tautoko ngā rōpū tohutohu mātanga ki te aroturuki me te aromatawai i te haumaru me te whai hua o ngā kano ārai mate KOWHEORI-19
Before February 2021, the Government had access to various groups that provided independent advice on medicines or vaccines.294 New Zealand advisory groups included the Medicines Assessment Advisory Committee, the Medicines Adverse Reactions Committee295, and international groups such as committees of the World Health Organization and the International Coalition of Medicines Regulatory Authorities.296
In February 2021, the Government established two new independent expert vaccine advisory groups in response to the COVID-19 pandemic. The COVID-19 Vaccine Technical Advisory Group had a focus on technical aspects of COVID-19 vaccines, and the COVID-19 Vaccine Independent Safety Monitoring Board had a focus on monitoring the safety of vaccines. Collectively, these groups had multidisciplinary expertise including in vaccinations, immunology, pharmacology, virology, clinical medicine, biostatistics, microbiology, and Māori and Pacific health. The Ministry of Health provided support to these advisory groups.
In February 2021, the Ministry of Health established the COVID-19 Vaccine Technical Advisory Group to provide science advice and recommendations on the use of COVID-19 vaccinations in the National Immunisation Programme, including on managing risks of harm. The Group received updates from the Centre for Adverse Reactions Monitoring and other groups tracking vaccine safety in New Zealand.
The Ministry of Health's Science and Technical Advisory Group supported the COVID 19 Vaccine Technical Advisory Group meetings throughout 2021 and 2022.297 The Ministry's Group produced fortnightly reports for the latter on the effectiveness and safety of the four COVID-19 vaccines in New Zealand's portfolio. It regularly (sometimes daily) searched for information from journal pre-prints, publications, international regulatory websites, infectious disease mailing lists, external COVID-19 data collation services, and news media sites. It produced daily summaries for senior Ministry of Health staff (including the Director-General).298
Also in February 2021, the Ministry of Health established the COVID-19 Vaccine Independent Safety Monitoring Board to provide expert advice on the safety of the COVID-19 vaccines. The Director-General of Health appointed the Chair, who subsequently appointed further members of the Board in accordance with World Health Organization guidance for independent groups of this nature. The Board met regularly throughout 2021 and 2022.299
Medsafe and other Ministry of Health staff attended the Board meetings as technical experts.300 The Board was able to call in additional expertise to help interpret the data around specific safety topics.301 The chairs of the Board and of the COVID-19 Vaccine Technical Advisory Group sometimes attended each other's meetings as ex-officio members.302
The Centre for Adverse Reactions Monitoring, the Immunisation Programme or Medsafe referred potential safety signals (including all reported deaths following vaccination) to the Board for review and expert opinion. The Board also considered international reports of adverse events and on actions taken by other regulators, and information on the background rates and pathophysiology of adverse events. If the Board agreed that there was a clear safety signal, it recommended action to Medsafe and the Immunisation Programme, including directly with the pharmaceutical company and through communications with the health sector and vaccination providers.303 For more detail, see Appendix 1, section 1.4.
Around November 2020, the Ministry of Health established a workstream as part of the National Immunisation programme, eventually known as the Vaccine Safety Surveillance and Research Group, to manage and report on the safety and effectiveness of the COVID-19 vaccines.304 The Group acted as the COVID-19 Vaccine Independent Safety Monitoring Board secretariat and worked with Medsafe and the Centre for Adverse Reactions Monitoring to make sure that adverse events were spontaneously reported, including through setting up additional safety monitoring systems. The Group also provided operational support for pharmacovigilance in Pacific countries, commissioned research on experience with COVID-19 vaccines in New Zealand and set up the Post Vaccine Symptom Check.305
2.1.6.7 Encouraging the reporting of adverse events | Te akiaki ki te whāki i ngā pāpono kino
In February 2021, the Ministry added a form to the COVID-19 Immunisation Register to help vaccinators report any immediate adverse events, such as anaphylaxis.306 In March 2021, the Ministry deployed another new form for reporting adverse events following COVID-19 vaccines and publicised it widely through the Medsafe and the Health Navigator New Zealand websites, newsletters and through communications with organisations who raised concerns about the safety of vaccines.307 To enable assessors to see reports they considered could be significant, a list of medically significant 'critical terms' was compiled that triggered a report being sent to an assessor for review.308
The Ministry took steps to encourage reporting of adverse events following COVID-19 vaccination. The Immunisation Advisory Centre was contracted to train vaccinators, including on how to use the Register and reporting forms.309 The Ministry worked closely with Healthline to ensure callers were advised to report adverse events.
The Ministry of Health agreed with the Office of the Chief Coroner to share information about coronial investigations into deaths potentially related to COVID-19. The Ministry's Vaccine Safety Surveillance and Research Group maintained a close relationship with the coroners to share information about cases of interest. As of 2025, there had been four coroners' findings in which a COVID-19 vaccine was implicated either as a direct or antecedent cause of the death.310 We discuss the reporting of vaccine-related deaths in more detail in Appendix 1, paragraphs 56–69.
In May 2021, the Ministry of Health issued the COVID-19 Vaccine Immunisation Programme Service Standards, which specified the mandatory, minimum requirements of the COVID-19 Vaccine Immunisation Programme. These aimed to ensure that vaccinators alerted consumers to potential side-effects of the vaccine and knew how to report them. The Ministry updated the Standards in September 2021.311
In August 2021, the Ministry of Health set up the Post Vaccine Symptom Check managed out of the National Immunisation Programme to help identify the rate of common, non-serious adverse events after vaccination. The Check administered an automated text-based survey sent to people's mobile phones, initially to a sample of adults recently vaccinated with Comirnaty. It asked about their vaccination experience, any early-onset adverse reactions, and any impacts on their daily life. The Ministry told us that the aim of the Check was to give the public confidence in the safety of the vaccines, through increasing transparency and communicating consumers' vaccination experience. It published the results on Medsafe's website.312
2.1.6.8 Building capacity and capability to assess reports of adverse events | Te whakapakari i te āheinga me te pūkenga hei aromatawai i ngā whāki pāpono kino
The Ministry of Health received advice that the number of reports resulting from vaccinating most of New Zealand's population might overwhelm the Centre for Adverse Reactions Monitoring database.313 It initiated constructing a new database with greater capacity and linked to the immunisation register to provide accurate information about vaccinations.314 This database was in place by March 2021.315
Part of Medsafe's process for assessing reports of adverse events includes comparing ordinary rates of events to observed rates following vaccination and to any specific trends or patterns that might indicate a vaccine safety concern. To identify safety signals from reports (and establish a causal relationship with vaccination), experts therefore need evidence of the usual rates of adverse events in the population (see section 2.1.6.3 and Appendix 1).316 In January 2021, the Ministry of Health created a list of adverse events that they were particularly interested in monitoring.317
Also in early 2021, the Ministry commissioned the University of Auckland to conduct a study to calculate background rates of adverse events relevant to COVID-19 vaccines in New Zealand (the SAFE study).318 The study assessed the background occurrence, between 2014–2019, of more than 30 types of adverse events of special interest. It compared this with data from 2020 to 2022 to identify if any events occurred at higher frequency after the vaccine rollout. The study aligned with the European Medicines Agency global protocol, enabling direct data comparisons and knowledge-sharing.319 The University of Auckland's report was finalised in December 2021 and provided to the Ministry of Health and Medsafe on 3 March 2022.320
The Ministry of Health used this research, combined with COVID-19 vaccination data linked to public hospital records, to identify whether people who had received the Comirnaty vaccine experienced specified adverse events at a different rate to the background rate.321
2.1.6.9 Systems for communicating about vaccine safety and cross-agency collaboration | Ngā pūnaha mō te whakawhiti kōrero mō te haumaru o ngā kano ārai mate me te mahi ngātahi ā-tari
The Ministry of Health communicated information about Medsafe's approval process and decision-making, and activities of the Centre for Adverse Reactions Monitoring through Medsafe's website. The website began providing information on COVID-19 from November 2020, including a dedicated vaccine safety monitoring webpage with information on reporting processes and monitoring.322 The Ministry of Health also took steps to encourage reporting (as outlined above), published results from its active surveillance methods and contributed to communication materials, including the Immunisation Handbook, to support awareness of vaccine safety monitoring.323
Other government agencies also had a role in considering and responding to harm caused by the COVID-19 vaccines, including the Accident Compensation Corporation (ACC). As of January 2025, ACC had accepted 1,740 claims for injuries related to the COVID-19 vaccines (out of 4,318 claims), five of which were for a fatal injury related to a COVID-19 vaccine.324 As required by statute, ACC reported three 'risks of harm', all related to errors in the administration of the COVID-19 vaccine.325 ACC was involved in the design of the Immunisation Programme. Although it was not involved in managing or encouraging reporting of adverse events,326 ACC notified all vaccine injury claims and the outcomes of its assessment to Medsafe, who captured them in the Centre for Adverse Reactions Monitoring's database and the signal detection process.
The Health and Disability Commissioner also received information about vaccine injuries within its complaint jurisdiction. The Commissioner referred five complaints about possible adverse reactions to the Ministry of Health to consider further and released one decision which we discuss later in section 2.1.9.1.327 The Health Quality and Safety Commission has a role in collecting data on morbidity and mortality but did not undertake any work specific to the vaccines during the pandemic.328
2.1.7 Responding to the emerging risk of myocarditis and pericarditis | Te urupare ki te tūraru e puta ake ana o te myocarditis me te pericarditis
During 2021, the heart conditions myocarditis and pericarditis329 emerged as serious but very rare side effects of mRNA vaccines, which international clinical trials of the Comirnaty vaccines had not identified. Although the risk of myocarditis and pericarditis was much greater from the virus itself, a number of submitters expressed concern that the New Zealand Government did not adequately convey these risks to the public.330 In this section, we outline how Medsafe and other health agencies monitored, confirmed and communicated the emerging risk during 2021 and how the Government responded in decisions to use Comirnaty in the National Immunisation Programme.
2.1.7.1 Medsafe promptly identified and notified myocarditis as a safety signal | I tautuhi wawe a Medsafe, ā, i whāki hoki i te myocarditis hei tohu haumaru
At a meeting of the International Coalition of Medicines Regulatory Authorities on 17 February 2021, Medsafe first became aware of the potential risk of myocarditis from using mRNA vaccines.331 Medsafe assessed there was then very little evidence to confirm the risk.
Medsafe received the first New Zealand report of possible vaccine-related myocarditis on 20 April.332 On 27 April, Medsafe began a signal investigation and on 29 April, the Covid-19 Vaccine Independent Safety Monitoring Board recommended monitoring this potential safety signal and establishing the background rate for myocarditis.333 The Centre for Adverse Reactions Monitoring received two further reports of myocarditis cases in New Zealand about 9 and 18 May.334
Medsafe first notified and began reviewing myocarditis as a safety signal in its weekly safety report on 26 May.335 The United States Centers for Disease Control and Prevention and the World Health Organization's vaccine safety committee had shortly before taken similar action in communicating the possible myocarditis risk and the need for further monitoring.336
On 27 May, the Covid-19 Vaccine Independent Safety Monitoring Board considered the two reports of myocarditis and a possible link with the Comirnaty vaccine. The Board recommended Medsafe issue an official monitoring communication for the myocarditis signal to encourage reporting of cases and so help Medsafe assess the possible link.337
Having considered the two reports of myocarditis in New Zealand, international developments, and the Board's recommendation, Medsafe published a monitoring communication on its website on 9 June.338 The communication identified myocarditis as a potential adverse reaction to Comirnaty and encouraged further reports. It advised:
Patients should NOT decline vaccination subject to a monitoring communication. If you have any concerns with your vaccination, please contact your healthcare professional. A monitoring communication does not mean that the vaccine, medicine or medical device causes an adverse reaction. [...] The benefits of the Comirnaty vaccine still outweigh the risks.
On the same day, the Ministry of Health advised subscribers to its COVID-19 Vaccine Update newsletter of the monitoring communication.339
2.1.7.2 The Government, in June 2021, delayed a decision on using Comirnaty for young people aged 12 to 15 years | I Hune 2021, i whakaroatia e te Kāwanatanga te whakatau mō te whakamahi i te Comirnaty mō ngā rangatahi 12–15 tau
On 21 June 2021, Medsafe extended provisional approval of Comirnaty for young people aged 12 to 15 years.340 However, because Medsafe had by this stage identified myocarditis as a potential adverse reaction to Comirnaty, on 28 June, Cabinet agreed with the advice of the COVID-19 Vaccine Technical Advisory Group to delay a decision on using Comirnaty for people in this age group, until further advice was provided on the risk of myocarditis.341 The Group advised that limited data made it hard to assess the relative risks and benefits of Comirnaty for this group, and that because COVID-19 case numbers among this group were low and vaccine supply was constrained, its use in this age group could be deferred.342
2.1.7.3 Medsafe confirmed myocarditis and pericarditis as new but rare side-effects of the Comirnaty vaccine in late July 2021 | I te mutunga o Hūrae 2021, i whakatūturu a Medsafe ko te myocarditis me te pericarditis he pānga taha hou, ahakoa he onge, o te kano ārai mate Comirnaty
On 9 July, the World Health Organization's Global Advisory Committee on Vaccine Safety reported a 'plausible causal relationship' between COVID-19 mRNA vaccines and myocarditis and pericarditis based on reports, including from the European Medicines Agency.343 Both the Agency and the United States Food and Drug Administration had communicated with healthcare professionals and the public advising that vaccinated people should seek medical attention if they developed symptoms suggestive of myocarditis or pericarditis (such as chest pain, shortness of breath or palpitations). The World Health Organization noted that myocarditis and pericarditis following vaccination was generally mild and responded well to conservative treatment (rest and anti inflammatory medication).
Medsafe continued to monitor the myocarditis safety signal over the period to 21 July. It found that reactions were more common following the second dose of the vaccine and that rates of myocarditis were higher in young males than expected.344 It published a further safety report and COVID-19 Vaccine Updates, noting the risk of myocarditis and that it was monitoring the situation.345 Staff from Medsafe, the Covid-19 Vaccine Independent Safety Monitoring Board, the COVID-19 Vaccine Technical Advisory Group and the Immunisation Programme met several times to discuss the risk, including how it should be factored in to the vaccine rollout.346
On 21 July, Medsafe published an alert communication on its website, confirming myocarditis and pericarditis as established but rare side effects of the Comirnaty vaccine.347 It instructed healthcare professionals and vaccinators to inform consumers of those risks and their symptoms.348 It advised that the benefits of vaccination with Comirnaty continued to outweigh the risks.
On the same day, the Ministry of Health issued a COVID-19 Vaccine Update identifying the risk of myocarditis as a rare adverse reaction to the Comirnaty vaccine. The Director-General drew attention to Medsafe's alert communication in a press conference. He advised that vaccinated people should seek immediate attention if they experienced new onset of chest pain or shortness of breath.349
On 2 August, Medsafe published an updated datasheet for Comirnaty.350 The datasheet told healthcare professionals to be alert to the signs and symptoms of myocarditis and pericarditis, and to tell consumers to seek immediate medical assistance if they developed those signs or symptoms. Medsafe also updated the consumer medicine information sheet on its website to include myocarditis and pericarditis as potential side effects.351 In late July, the Immunisation Advisory Centre updated its website to refer to myocarditis in the FAQs related to COVID-19 vaccine safety information.352
2.1.7.4 The Government considered emerging risks with Comirnaty and concluded that they did not outweigh the benefits | I āta whakaaro te Kāwanatanga ki ngā tūraru hou o Comirnaty, ā, i whakatau kāore aua tūraru e nui ake i ngā painga
On 21 July 2021, the day the alert communication was published, the COVID-19 Vaccine Technical Advisory Group gave comprehensive advice to the Director-General of Health about the myocarditis and pericarditis risk, after receiving guidance from a paediatric cardiologist.353
The Advisory Group noted that while New Zealand data did not currently indicate an association between Comirnaty and myocarditis, emerging data from overseas did, and the risk was higher following the second dose and in males and younger groups. It recommended that people aged 16 to 29 years receive a second dose of Comirnaty at least eight weeks after the first dose because this could reduce the frequency of some side effects.354 If people experienced myocarditis or pericarditis after a first dose, they should not have a second dose, unless they decided to do so after discussion with their healthcare provider. People in the higher-risk age group already under regular review by a cardiologist should also consult their healthcare team before being vaccinated. The Group noted that Medsafe and international regulators all agreed that the benefits of Comirnaty continued to outweigh the risks at an individual level.
On 22 July 2021, the Director-General of Health passed the advice of the COVID-19 Vaccine Technical Advisory Group to the Prime Minister and other relevant ministers.355 In doing so, the Director-General noted the need for the Government's communications to respond to growing public awareness of the risk of myocarditis and pericarditis. He noted that younger individuals were already more vaccine-hesitant than older groups and communications would need to be clear that the benefits of the Comirnaty vaccine outweighed the risks. He also noted that Medsafe and overseas regulators had approved it as safe and effective.
At a meeting on 27 July 2021, the Advisory Group again discussed the recommended dosing interval for people under 30 years. The Group discussed that while an eight-week interval was recommended for this age group, administering the second dose between six and 12 weeks after the first was acceptable, and that the exact timing was a programming decision.356 A follow-up meeting of the Group on 3 August 2021 confirmed that the Director-General of Health had accepted the recommendation to extend the dosing interval and an implementation plan was forthcoming.357
On 30 July 2021, the Director-General of Health received further advice from the COVID-19 Vaccine Technical Advisory Group about using Comirnaty for people aged 12 to 15 years.358 The advice recommended a limited rollout among that age group to those at particularly high risk of severe illness or death if they contracted COVID-19. This advice was ultimately superseded (see section 2.1.7.6).
The Government continued to use Comirnaty as its primary COVID-19 vaccine for the remainder of the period covered by our Inquiry. All advice was to the effect that, after considering the risk of myocarditis and pericarditis, Comirnaty continued to demonstrate a strong safety and efficacy profile and its use was instrumental in safeguarding public health during the pandemic.
2.1.7.5 The Ministry of Health communicated further after the first vaccine-related myocarditis death in August 2021 | I tuku kōrero anō te Manatū Hauora, i muri i te matenga tuatahi e pā ana ki te myocarditis nā te kano ārai mate, i Ākuhata 2021
On 2 August 2021, the Centre for Adverse Reactions Monitoring received the first report of potential vaccine-related myocarditis with a fatal outcome. On 9 August 2021, the Covid-19 Vaccine Independent Safety Monitoring Board agreed that the vaccine was the probable cause. On 13 August and 23 August 2021, the Board recommended that the National Immunisation Programme communicate to raise awareness of myocarditis as a risk.359
The Ministry of Health continued to communicate with the public and health professionals about the risk of myocarditis and pericarditis through:360
- COVID-19 Vaccine Updates on 27 August 2021 and 30 August 2021
- a media release on 30 August 2021, which was circulated widely by both local and international media
- media interviews on 30 August 2021 with the Ministry, Prime Minister, Director-General and the Chair of the COVID-19 Vaccine Independent Safety Monitoring Board
- a meeting with stakeholders including the Royal New Zealand College of General Practitioners, the Immunisation Advisory Centre and District Health Boards.
2.1.7.6 The Government extended the use of Comirnaty to young people over the age of 12 years in August 2021 and later to children aged 5–11 years | I Ākuhata 2021, i whakawhānui te Kāwanatanga i te whakamahi i te Comirnaty ki ngā rangatahi neke atu i te 12 tau, ā, nō muri mai ki ngā tamariki 5–11 tau
On 12 August 2021, the Government announced that the standard interval between Comirnaty doses would be increased for everyone, from three to six weeks.361 This was reported as being due to the extended duration conferring an equally robust immune response without any additional safety concerns, while also reducing the likelihood of myocarditis and pericarditis.362 With the interval lengthened, more people could receive an initial dose from the existing supply of vaccines.
On 16 August, Cabinet accepted an updated recommendation from the COVID-19 Vaccine Technical Advisory Group to extend the use of Comirnaty to all young people over the age of 12 years.363 The Minister for COVID-19 Response pointed to growing international evidence of the effectiveness of the vaccine for this age group and noted that the Delta variant of COVID-19 had increased transmissibility.364 On 17 August, New Zealand went into lockdown, after community cases (including with the Delta variant) were detected for the first time in 170 days.365
On 20 December 2021, on the recommendation of the Minister for COVID-19 Response, Cabinet agreed to use the paediatric Comirnaty vaccine for children aged 5 to 11 years in the national immunisation programme.366
When announcing the decision to approve the paediatric vaccine, the Minister noted that myocarditis and pericarditis were very rare side effects of the Comirnaty vaccine, but no cases had been reported in this age group in clinical trials. He said that the COVID-19 Vaccine Technical Advisory Group and Medsafe would continue to monitor safety and people in all age groups should be aware of the symptoms if they were vaccinated.367
2.1.7.7 The Government published further advice on the risk of myocarditis and pericarditis | I whakaputa kōrero ārahi anō te Kāwanatanga mō ngā tūraru o te myocarditis me te pericarditis
From September 2021, the Ministry of Health, Medsafe, and the Immunisation Advisory Centre continued to publish numerous updates, reminders and alerts about myocarditis and pericarditis and the signs and symptoms to look out for.368 The Centre outlined the risk of myocarditis and pericarditis in a number of immunisation programme materials sent to vaccinators and consumers.369
On 15 December 2021, the Ministry of Health asked District Health Board chief executives to distribute a letter to local providers. The letter requested that they make those vaccinated with Comirnaty aware of the possible symptoms of myocarditis or pericarditis and how to seek medical advice.370 Further communications and reminders occurred in early 2022.
Despite these communications, guidance was not always explicit about the need to discuss the risk of myocarditis and pericarditis, including before vaccinating as part of the informed consent process. This was highlighted by the Health and Disability Commissioner when she reviewed the information given to a person who subsequently died from myocarditis.371 The Ministry of Health's September 2021 Operating Guidelines for District Health Boards and Providers is an example of guidance that mentioned inflammation of the heart as a rare side effect but did not recommend it be discussed with patients before administering the vaccine.
The Immunisation Advisory Centre's guidance from September 2021 recommended vaccinators tell people of the need to seek medical advice if they experienced chest pain, shortness of breath, or palpitations.372 Yet, not until updates in February 2023 and July 2022 respectively, did either of these communications explicitly require vaccinators to discuss the risk of myocarditis and pericarditis as part of the informed consent process. We saw evidence that vaccinators sometimes did not give information on myocarditis and pericarditis because they did not want to worry people or felt pressured with time, and that brochures on myocarditis and pericarditis were not always passed onto consumers.373
2.1.8 Decisions resulted in a large increase in reports of adverse events and increased capacity to assess them | Nā aua whakatau i tino piki ake ai ngā whāki mō ngā pāpono kino, ā, i piki hoki te āheinga ki te aromatawai i aua whāki
The Ministry of Health's strategy was to encourage more reports of vaccine-related adverse events and increase the capacity to receive and assess them. As a result, the Centre for Adverse Reactions Monitoring database has greatly increased capacity to handle large numbers of reports and through encouragement and better information, people are now more aware of and familiar with reporting adverse events. Both before and during the pandemic, New Zealand had one of the highest rates of voluntary reporting per capita in the world.374
Up until the end of November 2022, the Centre for Adverse Reactions Monitoring received almost 64,000 reports of adverse events following administration of almost 12.5 million doses of COVID-19 vaccines. Over 60,000 of those were for the Comirnaty adult vaccine reflecting that many more doses of that vaccine were administered than of other vaccines. The most frequent adverse events reported for Comirnaty were headache, injection site pain, lethargy, lymphadenopathy, pyrexia, influenza-like illness, nausea, dizziness, chest discomfort and swelling. Many of those reactions were minor and/or known risks associated with the vaccines and, while they continued to be monitored, the COVID-19 Vaccine Independent Safety Monitoring Board considered they did not require further investigation.375
On referral from Medsafe, the expert Covid-19 Vaccine Independent Safety Monitoring Board assessed 784 serious reports (defined in section 2.1.6.3) using scientific evidence on background rates to do so.376 The Board identified 25 possible new safety signals (24 for Comirnaty, one for Novavax).377 Three of those signals (myocarditis and pericarditis, anaphylaxis and Bell's Palsy) were established as being associated with the Comirnaty vaccine. The Board made 40 recommendations to Medsafe or the National Immunisation Programme to continue routine monitoring, occasionally also recommending additional communication to health professionals.
We saw evidence that harmful and inaccurate information emerged about the vaccines' safety after they began to be used, alongside genuine concerns. Appendix 1 addresses some of the most prevalent concerns. Because of those concerns, some New Zealanders refused the COVID-19 vaccines and lost trust in government.
2.1.9 Key decisions: post-approval vaccine safety – our assessment | Ngā whakatau matua: te haumaru o te kano ārai mate i muri i te whakaaetanga – tā mātou aromatawai
2.1.9.1 The system to monitor the safety of COVID-19 vaccines was comprehensive and responsive | He whānui, he urupare hoki te pūnaha hei aroturuki i te haumaru o ngā kano ārai mate KOWHEORI-19
The Ministry of Health and other decision-makers anticipated the need for a robust and comprehensive system to monitor the safety of COVID-19 vaccines. They were well-informed by expert advice about past practice and the strengths and weaknesses in the system and took steps to ensure the system could cope with increased demand and respond quickly to emerging risk (such as myocarditis and pericarditis).
This expert advice was also clear about how a strong, responsive and transparent vaccine safety monitoring system would support trust and confidence in the immunisation programme. Such trust was essential to widespread acceptance of the benefits of vaccination.
New Zealand's system to monitor vaccine safety was linked to, and well aligned with, those of international medicines regulators. New Zealand was, as a result, well placed to act quickly when new concerns were raised. The actions taken in response, and advice to decision-makers, the health sector and the public, was prompt and accurate.
Better coordinated communications to providers on emerging risk would have supported providers providing clearer information on risks to consumers
In 2020, both the World Health Organization and Medsafe's advisors recommended that the system to monitor COVID-19 vaccine safety include effective communication. As the Health and Disability Commissioner noted, to support the informed consent process between health care providers and consumers, the Ministry of Health needed to ensure providers were alerted to any new and emerging side effects appropriately and effectively and in a timely matter.378
The evidence shows that the Government made numerous efforts to communicate vaccine safety issues, including the emerging risk of myocarditis and pericarditis. Key immunisation guidance was updated by October 2021.379 However, communications were disseminated from several sources. Although the advice was consistent about the nature of the risk, as the Health and Disability Commissioner noted, key messages were not always explicit or emphasised or were lost in the volume of information providers were receiving from several official sources.380
The provision of information about vaccines and the obtaining of informed consent is the responsibility of vaccinators and other health providers.381 Even so, the Ministry of Health has an important role in ensuring vaccinators and other providers have the information they need about vaccines, to support their discussions with consumers. This is especially important when inducting an entirely new vaccination workforce, as happened during the COVID-19 pandemic.
The Health and Disability Commissioner asked Phase One of the Inquiry to consider the feasibility of a centralised portal for all information for providers. Phase One found that '[w]hile the frequency and changing content of these updates [on the potential risk of myocarditis following vaccination] reflected a desire to communicate the most current evidence, it was challenging for people to keep on top of and process this information.382 Phase One agreed with the Health and Disability Commissioner on 'the desirability of having stronger mechanisms for providing clear and consistent advice on vaccine risks'.383
We concur with that assessment. We acknowledge and endorse the steps taken to communicate vaccine safety and emerging risks. However, stronger coordination of that messaging would have been beneficial.
2.1.10 Summary assessments | Ngā whakarāpopototanga aromatawai
Key decisions on vaccine approvals and monitoring vaccine safety were led out of the Ministry of Health, particularly Medsafe. The decisions were based on extensive advice from medical and scientific experts, who closely monitored evolving international experience around developing, approving and using COVID-19 vaccines.
Decisions carefully balanced the benefits of novel COVID-19 vaccines for the health and safety of New Zealanders, against potential harms from their approval. Decision-makers took extensive steps to mitigate risks through strengthening systems for monitoring the safety of vaccines once they were in use. At the same time, the Government viewed widespread acceptance and use of vaccines as a key means to end restrictive public health measures and so achieve the economic, social and educational benefits that this would bring.
Decisions carefully balanced the benefits of novel COVID-19 vaccines for the health and safety of New Zealanders against potential harms from their approval.
Decision-makers were fully aware of the need to maintain public acceptance of vaccines. However, they did not anticipate the extent to which concerns about vaccine safety would emerge. Communications around risks from COVID-19 vaccines were extensive and followed established procedures. Even so, shortfalls in clarity and effectiveness existed in terms of what providers heard about risks and, in turn, communicated to consumers.
118 World Health Organization, 'Vaccines and immunization: What is vaccination?' (22 October 2025), https://www.who.int/news-room/questions-and-answers/item/vaccines-and-immunization-what-is-vaccination
119 World Health Organization, 'Vaccines and immunization', https://www.who.int/health-topics/vaccines-and-immunization
120 Herd immunity, in the context of vaccination, is when enough people in a group or area have become immune (protected) against a virus to make it difficult for it to spread. It can happen through people developing immunity through catching the infection, or through vaccination. Health New Zealand | Te Whatu Ora, 'Immunisation Handbook 2025, Version 7' (17 November 2025), https://www.tewhatuora.govt.nz/for-health-professionals/clinical-guidance/immunisation-handbook, p 17
121 Shahul H Ebrahim, Qanta A Ahmed, Ernesto Gozzer, Patricia Schlagenhauf and Ziad A Memish, 'Covid-19 and community mitigation strategies in a pandemic', British Medical Journal, vol 368 (17 March 2020), https://www.bmj.com/content/368/bmj.m1066
122 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
123 Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand: Pharmacovigilance, ed 3.0 (March 2024), https://www.medsafe.govt.nz/Regulatory/Guideline/GRTPNZ/part-8-pharmacovigilanceEdition3March2024.pdf, p 8
124 COVID-19 Vaccine Independent Safety Monitoring Board, Final report 2022: COVID-19 Vaccine Independent Safety Monitoring Board (3 July 2023), https://www.tewhatuora.govt.nz/publications/final-report-of-the-covid-19-vaccine-independent-safety-monitoring-board-cv-ismb, p 3
125 COVID-19 Vaccine Independent Safety Monitoring Board, Final report 2022: COVID-19 Vaccine Independent Safety Monitoring Board (3 July 2023), https://www.tewhatuora.govt.nz/publications/final-report-of-the-covid-19-vaccine-independent-safety-monitoring-board-cv-ismb, p 3
126 New Zealand was one of the founding members of the World Health Organization International Drug Monitoring Programme when it was established in 1968. Desiree L. Kunac, Mira Harrison-Woolrych and Michael V. Tatley, 'Pharmacovigilance in New Zealand: the role of the New Zealand Pharmacovigilance Centre in facilitating safer medicines use', New Zealand Medical Journal, vol 121:1283 (3 October 2008), https://pubmed.ncbi.nlm.nih.gov/18841188/, p 76
127 Early antivirals used to treat COVID-19 were repurposed from other diseases and had limited or unclear evidence of efficacy against COVID-19.
128 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2 Section 7.2.1, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part-two/7-2-what-happened;
Ministry of Business, Innovation and Employment, Briefing: COVID-19 Vaccine Strategy – Purchasing strategy and funding envelope (10 July 2020), https://covid19.govt.nz/assets/Proactive-Releases/proactive-release-2020-october/HR28-2021-0139-COVID-19-Vaccine-Strategy-Purchasing-Strategy-and-funding-en....pdf, p 4
129 New Zealand did not use the Moderna (Spikevax) COVID-19 vaccine.
130 Environmental Protection Authority, 'The spike protein and the Pfizer vaccine' (11 May 2021), https://www.epa.govt.nz/community-involvement/science-at-work/sars-cov-2-vaccines-and-the-hsno-act
131 Medsafe, 'mRNA Vaccines' (revised 15 April 2021), https://www.medsafe.govt.nz/COVID-19/mRNA-vaccines.asp; World Health Organization, COVID-19 Vaccines: Safety Surveillance Manual, second edition (2021), https://iris.who.int/server/api/core/bitstreams/404bd389-32fb-4c0d-a2cb-1e4899232ea5/content, Section 6.1 p 5
132 Medsafe, 'Viral Vector Vaccines' (revised 7 July 2021), https://www.medsafe.govt.nz/COVID-19/viral-vector-vaccines.asp;
World Health Organization, COVID-19 Vaccines: Safety Surveillance Manual, second edition (2021), https://iris.who.int/server/api/core/bitstreams/404bd389-32fb-4c0d-a2cb-1e4899232ea5/content, Section 6.1 p 5
133 World Health Organization, COVID-19-19 Vaccine Explainer: COVID-19 Vaccine (recombinant, adjuvanted), NVX-CoV2373, COVOVAX™, NUVAXOVID™ (21 January 2022), https://www.who.int/docs/default-source/coronaviruse/4-feb_22015_novavax_vaccine-explainer_for_web.pdf, p 1
134 European Medicines Agency, 'COVID-19 vaccines: development, evaluation, approval and monitoring', https://www.ema.europa.eu/en/human-regulatory-overview/public-health-threats/coronavirus-disease-covid-19/covid-19-public-health-emergency-international-concern-2020-23/covid-19-vaccines-development-evaluation-approval-monitoring
135 'In vitro' refers to processes or reactions occurring in a controlled environment outside a living organism, such as in a test tube or culture dish.
136 'In vivo' refers to studies taking place in a living organism.
137 Ulrich Kalinke, Dan H. Barouch, Ruben Rizzi, Eleni Lagkadinou, Ozlem Turci, Shanti Pather and Pieter
Neels, ‘Clinical development and approval of COVID-19 vaccines’, Expert Review of Vaccines, vol 21:5 (2022),
https://doi.org/10.1080/14760584.2022.2042257, pp 609–619
138 Ann J. Barbier, Allen Yujie Jiang, Peng Zhang, Richard Wooster and Daniel G. Anderson, ‘The clinical
progress of mRNA vaccines and immunotherapies’ Nature Biotechnology, vol 40:6 (9 May 2022),
https://doi.org/10.1038/s41587-022-01294-2, pp 840–854
139 Norbert Pardi, Michael J. Hogan, Frederick W. Porter and Drew Weissman, ‘mRNA vaccines – a new
era in vaccinology’, Nature Reviews Drug Discovery, vol 17 (12 January 2018), https://doi.org/10.1038/
nrd.2017.243, pp 261–279
140 British Society for Immunology, ‘How have COVID-19-19 vaccines been developed so fast?’,
https://www.immunology.org/public-information/vaccine-resources/covid-19/covid-19-vaccine-
infographics/speed-of-development
141 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, engagement with
Professor David Murdoch (5 June 2025)
142 That strategy – later known as the COVID-19 Immunisation Strategy and Programme – was developed
further and endorsed by Cabinet in December 2020.
Cabinet Minute, CAB-20-MIN-0229.01, COVID-19 Vaccine Strategy (18 May 2020), https://fyi.org.nz/
request/25063/response/96453/attach/5/Release%20Cabinet%20Papers.pdf, pp 1–3
Cabinet Minute, CAB-20-MIN-0509, Update on the COVID-19 Immunisation Strategy and Programme
(7 December 2020), https://www.health.govt.nz/system/files/2021-05/cab-20-min-0509_0.pdf, pp 4–9
143 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, interview with
Rt. Hon. Dame Jacinda Ardern (30 July 2025)
144 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, interview with
Hon. Grant Robertson (28 July 2025)
145 Cabinet Minute, CAB-20-MIN-0229.01, COVID-19 Vaccine Strategy (18 May 2020), https://fyi.org.nz/
request/25063/response/96453/attach/5/Release%20Cabinet%20Papers.pdf, pp 1–3
NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, interview with
Hon. Grant Robertson (28 July 2025)
NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, interview with Rt. Hon. Dame Jacinda Ardern (30 July 2025)
146 Cabinet Paper, COVID-19 Vaccine Strategy: Update on vaccine purchasing [n.d]
147 Cabinet Paper, COVID-19 Vaccine Strategy (June 2020), https://covid19.govt.nz/assets/Proactive-Releases/proactive-release-2020-june/PAPER-COVID-19-Vaccine-Strategy.pdf, p 10
148 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two (2026), Pandemic
Perspectives, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/phase-two/
pandemic-perspectives
149 World Health Organization, 'The World Health Organization Programme for International Drug Monitoring, Programme for International Drug Monitoring', https://www.who.int/teams/regulation-prequalification/regulation-and-safety/pharmacovigilance/networks/pidm
150 Royal Commission of Inquiry (COVID-19 Lessons) Amendment Order (No 2) 2024, Schedule 2 cl 6,
https://www.legislation.govt.nz/regulation/public/2024/0177/latest/LMS984331.html
151 Royal Commission of Inquiry (COVID-19 Lessons) Amendment Order (No 2) 2024, Schedule 2 cl 6,
https://www.legislation.govt.nz/regulation/public/2024/0177/latest/LMS984331.html
152 Cabinet Minute, CAB-20-MIN-0382, COVID-19 Vaccine Strategy: Purchasing Strategy and Funding Options
(10 August 2020), https://covid19.govt.nz/assets/Proactive-Releases/proactive-release-2020-october/
HR02-CABINET-PAPER-AND-MINUTE-COVID-19-vaccine-strategy-10-August-2020.pdf, pp 17–20
153 Controller and Auditor-General, Preparations for the nationwide roll-out of the Covid-19 vaccine
(May 2021), https://oag.parliament.nz/2021/vaccines/docs/vaccines-roll-out.pdf, p 19
154 Cabinet Paper, February 2021 update on the COVID-19 Immunisation Strategy and Programme (February
2021), https://www.health.govt.nz/system/files/2021-05/february_2021_update_on_the_covid-19_
immunisation_strategy_and_programme.pdf
155 Hon. Dr Megan Woods and Hon. Chris Hipkins, ‘First COVID-19 vaccine purchase agreement signed’
(12 October 2020), https://www.beehive.govt.nz/release/first-covid-19-vaccine-purchase-agreement-signed
156 Ministry of Health, response to OIA request H202116355 (November 2021), https://fyi.org.nz/
request/17594/response/68208/attach/3/H202116355%20Response.pdf, p 2
157 While Medsafe later gave consent for a form of the vaccine to be given to children under five years old,
this decision was not made until 15 November 2023 and is therefore outside the scope of this Inquiry.
Medsafe, ‘COVID-19 Archive: Approval status of COVID-19 vaccine applications received by Medsafe’
(revised 9 June 2024), https://medsafe.govt.nz/COVID-19/status-of-applications.asp#comirnatyu5
158 Previously named COVID-19 Vaccine Janssen. Also known as the Johnson and Johnson vaccine in
some jurisdictions.
159 Medicines Act 1981, s 20, https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55054.html
160 Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/files/2021-10/h202106950_-_response.pdf, Document 1
Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf, Document 2
Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf, Document 3
161 The legislation refers to a ‘manufacturer’, ‘importer’ or ‘proprietor’. Medicines Act 1981, ss 21 and 23,
https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55451.html
162 Medicines Act 1981, s 21(2), https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55057.html
163 Medicines Act 1981, s 23, https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55061.html
164 Medicines Act 1981, s 23, https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55061.html
165 Nga Kaitiaki Tuku Iho Medical Action Society Incorporated v Minister of Health and others [2021] NZHC 1107
(18 May 2021)
166 Medicines Act 1981, s 22(1)(a), https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55060.html
167 Medicines Act 1981, s 22(1)(b), https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55060.html
168 Medicines Act 1981, ss 22(2) and (5), https://www.legislation.govt.nz/act/public/1981/0118/latest/DLM55060.html
169 Medsafe, ‘Medsafe’s Evaluation and Approval Process’ (revised 12 September 2019), https://www.
medsafe.govt.nz/Consumers/Safety-of-Medicines/Medsafe-Evaluation-Process.asp#Process
Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand, Part 2: Obtaining approval
for new and changed medicines and related products, ed 1.2 (October 2020), https://www.medsafe.govt.
nz/regulatory/Guideline/GRTPNZ/Part2.pdf
A datasheet is a document supplied by the vaccine manufacturer that sets out the key information
about how to use the medicine safely and effectively. It is primarily aimed at health practitioners and is
published by Medsafe.
170 The Medsafe website states that ‘the extent of clinical studies varies depending on the type of
medicine application’. Medsafe, ‘Medsafe’s Evaluation and Approval Process’ (revised 12 September
2019), https://www.medsafe.govt.nz/Consumers/Safety-of-Medicines/Medsafe-Evaluation-Process.
asp#Process
171 Such as guidelines published by the European Medicines Agency, the U.S. Food and Drug Administration,
Health Canada, the Therapeutic Goods Administration (Australia) and the International Council for
Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
172 Medsafe, ‘Medsafe’s Evaluation and Approval Process’ (revised 12 September 2019), https://www.
medsafe.govt.nz/Consumers/Safety-of-Medicines/Medsafe-Evaluation-Process.asp#Process
173 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
174 Annette King, ‘Meningococcal B vaccine approved’ (8 July 2004), https://www.beehive.govt.nz/release/meningococcal-b-vaccine-approved
175 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
176 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
Cabinet Paper, COVID-19 Vaccines Strategy – early progress (22 July 2020), https://covid19.govt.nz/assets/
Proactive-Releases/proactive-release-2020-october/HR40-Minute-and-Cabinet-Paper-COVID-19-vaccine
strategy-early-progress.pdf, p 2
177 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
178 The formal criteria for granting priority status are that (a) the medicine is indicated for the treatment or diagnosis of a serious, life-threatening or severely debilitating disease or condition, for which other treatment options are limited; or (b) the product is required to address an out-of-stock situation, or withdrawal of alternative medicines, and maintaining access to the treatment is essential. We have not identified any negative population impacts of this prioritisation.Ministry of Health, Memo: Priority Assessment Request (10 December 2020)
Medsafe, ‘COVID-19 Archive: Vaccine Evaluation and Approval Process’ (27 November 2020),
https://www.medsafe.govt.nz/COVID-19/vaccine-approval-process.asp
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
179 They attended weekly meetings with the Medsafe team, reviewed drafts of evaluation reports, and
were personally involved in decisions to seek further information from companies and from other
external experts.
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition
to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
180 Medsafe, ‘COVID-19 Archive: Vaccine Evaluation and Approval Process’ (27 November 2020),
https://www.medsafe.govt.nz/COVID-19/vaccine-approval-process.asp
181 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition
to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
182 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
183 Environmental Protection Authority, Decision: APP204176 (11 February 2021), https://www.epa.govt.nz/
assets/FileAPI/hsno-ar/APP204176/APP204176-Decision.pdf
184 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition
to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
New Zealand Gazette, 2021-go338, Provisional Consent to the Distribution of a New Medicine (3 February 2021), https://gazette.govt.nz/notice/id/2021-go338
185 Ministry of Health, Memo: Provisional consent to distribute medicines under section 23 of the
Medicines Act 1981 (03 February 2021)
New Zealand Gazette, 2021-go338, Provisional Consent to the Distribution of a New Medicine
(3 February 2021), https://gazette.govt.nz/notice/id/2021-go338
186 Ministry of Health, New Zealand Datasheet Comirnaty COVID-19 Vaccine (as at 3 February 2021)
187 Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/files/2021-10/h202106950_-_response.pdf
188 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
189 Adverse events are unintended, unexpected or unplanned occurrences that occur to participants in
clinical trials. They are not necessarily related to the administration of the investigational medicinal
product. Serious adverse events (such as death; life-threatening at the time of the event; resulting in
inpatient hospitalisation) were low and similar across vaccinated and control groups in the Comirnaty
clinical trial (0.6% and 0.5%, respectively). Published results report 1.1% severe adverse events; however
Medsafe’s evaluation reports 1.2% severe adverse events.
Fernando P. Polack and others., ‘Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine’, N Engl J Med,
Vol 383 (10 December 2020), https://www.nejm.org/doi/full/10.1056/NEJMoa2034577, pp 2603–2615
Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf, Document 10
190 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
191 Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/files/2021-10/h202106950_-_response.pdf
192 Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf
193 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
Ministry of Health, Medsafe COVID-19 Vaccine Advisory Group – Request for Advice (11 January 2020)
Ministry of Health, Medsafe COVID-19 Vaccine Advisory Group – Request for Advice (12 January 2021)
Ministry of Health, Medsafe COVID-19 Vaccine Advisory Group – Request for Advice (18 January 2021)
194 A Risk Management Plan outlines known and potential risks from the use of a medicine and describes
how sponsors plan to monitor and manage those risks after approval.
Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
195 The Medicines Adverse Reactions Committee is a committee established under section 8 of the Medicines
Act 1981. Its role is to advise the Minister and Medsafe on medicine safety issues primarily after approval
and supply of a new medicine.
Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
Medsafe, ‘Committees: Medicines Adverse Reactions Committee’ (revised 22 March 2016), https://www.
medsafe.govt.nz/profs/MARC/TermsofReference.asp
196 The Science and Technical Advisory Group was a technical group established by the Ministry’s COVID-19
response team.
197 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
198 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
199 Medsafe can refer matters to an appropriate committee under s 22(2) of the Medicines Act 1981 if it is
not satisfied that consent should be given.
200 Medsafe, ‘Summary of Recommendations from the 109th meeting of the Medicines Assessment Advisory
Committee held in Wellington on Tuesday 2 February 2021 at 9:30am’, https://www.medsafe.govt.nz/
committees/maac/Recommendation109-2February2021.htm
201 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
202 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to
Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
203 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
204 Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf, Document 15
205 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders (5 May 2021)
206 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
Ministry of Health, ‘COVID-19 vaccine update’ (3 February 2021), https://www.youtube.com/
watch?v=jz6zCaFSWYw
207 New Zealand Gazette, 2021-go2440, Provisional Consent to the Distribution of a New Medicine
(21 June 2021), https://gazette.govt.nz/notice/id/2021-go2440
Ministry of Health, Memo: Pfizer New Zealand’s application for consent of the Comirnaty 0.5 mg/ml,
for 12–15 year olds – decision required under section 20 or section 23 of the Medicines Act 1981
(18 June 2021)
208 Ministry of Health, Memo: Pfizer New Zealand’s application for consent of the Comirnaty 0.5 mg/ml,
for 12–15 year olds – decision required under section 20 or section 23 of the Medicines Act 1981
(18 June 2021)
209 New Zealand Gazette, 2021-go4666, Renewal of Provisional Consent to the Distribution of a Medicine
(28 October 2021), https://gazette.govt.nz/notice/id/2021-go4666
210 Immunisation Advisory Centre, ‘Provisional approval for Pfizer vaccine extended’ (20 October 2021),
https://www.immune.org.nz/news/provisional-approval-for-pfizer-vaccine-extended
211 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
New Zealand Gazette, Provisional Consent to the Distribution of a New Medicine (8 November 2021),
https://gazette.govt.nz/notice/id/2021-go4766
212 New Zealand Gazette, 2021-go5403, Provisional Consent to the Distribution of New Medicines
(16 December 2021), https://gazette.govt.nz/notice/id/2021-go5403
Ministry of Health, Memo: Pfizer New Zealand Limited’s application for consent of Comirnaty
suspension for injection 30 µg/0.3 ml (TTS0-10853/1) and Comirnaty concentrate for injection
10 µg/0.2 ml (TT50-1053/1 a) – decision required under section 23 of the Medicines Act 1981
and under regulation 52 of the Medicines Regulations 1984 (16 December 2021)
213 Ministry of Health, Memo: Pfizer New Zealand Limited’s application for consent of Comirnaty
suspension for injection 30 µg/0.3 ml (TTS0-10853/1) and Comirnaty concentrate for injection
10 µg/0.2 ml (TT50-1053/1 a) – decision required under section 23 of the Medicines Act 1981 and
under regulation 52 of the Medicines Regulations 1984 (16 December 2021)
214 This included the need for additional information on some quality issues, transmission, Māori and
Pacific children and those with prior COVID-19 infection and post-marketing surveillance data,
including on myocarditis and pericarditis, which was an adverse event of special interest at that time.
Medsafe, response to OIA request H202117877, https://www.health.govt.nz/system/files/2022-02/h202117877_response.pdf, Document 1, p 7
215 Medsafe, response to OIA request H202117877, https://www.health.govt.nz/system/files/2022-02/h202117877_response.pdf, Document 1, p 7
216 Medsafe, ‘Minutes of the 188th Medicines Adverse Reactions Committee Meeting’ (2 December 2021),
https://www.medsafe.govt.nz/profs/adverse/Minutes188.htm#3.2.3, Section 3.2.3
217 Ministry of Health, Affidavit of Christopher Mark James on behalf of the Respondents in Opposition
to Application for Interim Orders, CIV-2021-485-181 (5 May 2021)
218 Ministry of Health, Memo: Pfizer New Zealand Limited’s application for consent of Comirnaty
suspension for injection 30 µg/0.3 ml (TTS0-10853/1) and Comirnaty concentrate for injection
10 µg/0.2 ml (TT50-1053/1 a) – decision required under section 23 of the Medicines Act 1981 and
under regulation 52 of the Medicines Regulations 1984 (16 December 2021)
219 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
220 World Health Organization, ‘Statement of the WHO Global Advisory Committee on Vaccine Safety (GACVS) COVID-19 subcommittee on safety signals related to the AstraZeneca COVID-19 vaccine’
(19 March 2021), https://www.who.int/news/item/19-03-2021-statement-of-the-who-global-advisory
committee-on-vaccine-safety-(gacvs)-covid-19-subcommittee-on-safety-signals-related-to-the-astrazeneca-covid-19-vaccine
World Health Organization, ‘Statement of the WHO Global Advisory Committee on Vaccine Safety
(GACVS) COVID-19 subcommittee on reports of Guillain-Barrē syndrome (GBS) following adenovirus
vector COVID-19 vaccines’ (26 July 2021), https://www.who.int/news/item/26-07-2021-statement-of-the
who-gacvs-covid-19-subcommittee-on-gbs
221 Ministry of Health, Memo: Biocelect New Zealand Ltd’s application for consent of Nuvaxovid solution
for injection 10 µg/mL (TT50-10879) – decision required under section 23 of the Medicines Act 1981 and
under regulation 52 of the Medicines Regulations 1984 (3 February 2022)
Ministry of Health, Memo: Consent to distribute a new medicine under section 20 of the Medicines Act
1981 (13 July 2021)
Ministry of Health, Memo: AstraZeneca Limited’s application for consent of COVID-19 Vaccine
AstraZeneca 5x1010 VP/0.5mL suspension for injection (TT50-10877) – decision required under section 23
of the Medicines Act 1981 and under regulation 52 of the Medicines Regulations 1984 (22 July 2021)
Medsafe, ‘Summary of Recommendations from the 112th meeting of the Medicines Assessment Advisory
Committee held in Wellington on Tuesday 15 June 2021’, https://medsafe.govt.nz/committees/maac/Recommendation112-15July2021.htm
222 Ministry of Health, Memo: Biocelect New Zealand Ltd’s application for consent of Nuvaxovid solution
for injection 10 µg/mL (TT50-10879) – decision required under section 23 of the Medicines Act 1981
and under regulation 52 of the Medicines Regulations 1984 (3 February 2022)
Ministry of Health, Memo: Consent to distribute a new medicine under section 20 of the Medicines
Act 1981 (13 July 2021)
Ministry of Health, Memo: AstraZeneca Limited’s application for consent of COVID-19 Vaccine
AstraZeneca 5x1010 VP/0.5mL suspension for injection (TT50-10877) – decision required under
section 23 of the Medicines Act 1981 and under regulation 52 of the Medicines Regulations 1984
(22 July 2021)
223 New Zealand Gazette, 2021-go2704, Provisional Consent to the Distribution of a New Medicine
(7 July 2021), https://gazette.govt.nz/notice/id/2021-go2704
224 New Zealand Gazette, 2021-go3088, Provisional Consent to the Distribution of a New Medicine
(29 July 2021), https://gazette.govt.nz/notice/id/2021-go3088
225 New Zealand Gazette, 2022-go330, Provisional Consent to the Distribution of a New Medicine
(4 February 2022), https://gazette.govt.nz/notice/id/2022-go330
226 Ministry of Health, Datasheet: COVID-19 Vaccine Janssen: Ad26.COV2.S (8 November 2021)
Ministry of Health, Datasheet: COVID-19 Vaccine AstraZeneca (29 July 2021)
227 Nga Kaitiaki Tuku Iho Medical Action Society Incorporated v Minister of Health and others [2021] NZHC 1107
228 Medicines Act 1981, s 23(1) (as at 29 September 2020), https://www.legislation.govt.nz/act/public/1981/0118/81.0/DLM55061.html
229 Nga Kaitiaki Tuku Iho Medical Action Society Incorporated v Minister of Health and others [2021] NZHC 1107,
at para 69
230 Cabinet Minute, CAB-21-MIN-0170, Proposed Amendment to the Medicines Act 1981: Provisional Approvals
(17 May 2021), https://www.health.govt.nz/system/files/2022-08/cab-21-min-0170_minute.pdf, p 1
231 We acknowledge that some submitters to Phase Two take issue with the amendments to the Medicines
Act 1981. However, it is out of scope for the Inquiry to consider the operation of the general regulatory
system for vaccines. See Royal Commission of Inquiry (COVID-19 Lessons) Amendment Order (No 2) 2024,
Schedule 2 cl 6, https://www.legislation.govt.nz/regulation/public/2024/0177/latest/LMS984331.html
232 Medicines Amendment Act 2021, Schedule 1AA cl 1(1)(a), https://www.legislation.govt.nz/act/
public/2021/0016/latest/whole.html#LMS496452
233 MKD v Minister of Health [2022] NZHC 1997
234 MKD v Minister of Health [2022] NZHC 1997, at paras 48 and 54²³⁵ Ministry of Health, Brief of Evidence of Christine May Nolan (19 August 2022)
235 Nga Kaitiaki Tuku Iho Medical Action Society Incorporated v Minister of Health and others [2021] NZHC 1107,
at para 69
236 Therapeutic Goods Administration, Australian Public Assessment Report for BNT162b2 (mRNA) (January
2021), https://www.tga.gov.au/sites/default/files/auspar-bnt162b2-mrna-210125.pdf, Table 1, pp 10–11
237 Matthew P. Doogue, ‘A safe and effective drug?’, New Zealand Medical Journal, Vol 127:1404
(17 October 2014), https://nzmj.org.nz/media/pages/journal/vol-127-no-1404/a-safe-and-effective-drug/
f080d9f2aa- 1696478466/a-safe-and-effective-drug.pdf
238 As outlined, the risks and safety concerns associated with the AstraZeneca, Janssen and Novavax vaccines
were known to Medsafe on approval.
239 Cabinet Minute, CAB-20-MIN-0509, Update on the COVID-19 Immunisation Strategy and Programme
(7 December 2020), https://www.health.govt.nz/system/files/2021-05/cab-20-min-0509_0.pdf
240 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2
Section 7.1, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part
two/7-1-vaccination-introduction
241 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2 Section 7.3, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part
two/7-3-our-assessment
242 Controller and Auditor-General, Preparations for the nationwide roll-out of the Covid-19 vaccine
(May 2021), https://oag.parliament.nz/2021/vaccines
Waitangi Tribunal, Haumaru: the COVID-19 Priority Report (2023), https://www.waitangitribunal.govt.nz/assets/DOCUMENTS/Haumaru-W.pdf
243 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2
Section 7.3, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part
two/7-3-our-assessment
244 Use of a specific COVID-19 vaccine in the Government’s COVID-19 Immunisation Programme was
explicitly reliant on Medsafe having first given approval.
Cabinet Paper, CAB-21-MIN-0012, Pfizer recommendations for decision to use (9 February 2021),
https://www.health.govt.nz/system/files/2021-05/pfizer_recommendations_for_decision_to_use.pdf, p 3
245 Access for different population groups was governed by a sequencing framework that Cabinet agreed
to in early 2021.
Cabinet Paper, CAB-21-MIN-0012, Pfizer recommendations for decision to use (9 February 2021),
https://www.health.govt.nz/system/files/2021-05/pfizer_recommendations_for_decision_to_use.pdf
Cabinet Minute, CAB-21-MIN-0052, Updated Approach to the Sequencing Framework for COVID-19
Vaccines (8 March 2021), https://www.health.govt.nz/system/files/2021-05/cab-21-min-0052_minute.pdf
246 Cabinet Paper, CAB-21-MIN-0012, Pfizer recommendations for decision to use (9 February 2021),
https://www.health.govt.nz/system/files/2021-05/pfizer_recommendations_for_decision_to_use.pdf
Ministry of Health, Memo: Decision to use the Pfizer mRNA COVID-19 vaccine: COVID-19 Vaccine
Technical Advisory Group (CV TAG) recommendations (5 February 2021), https://www.tewhatuora.govt.
nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine-Technical-Advisory-Group-CV-TAG/
Decision-to-use-the-Pfizer-mRNA-COVID-19-vaccine.pdf
247 Although there had been no clinical data about the use of Comirnaty in immunocompromised, pregnant
and lactating women at the time Medsafe gave approval, advisors were comfortable that potential risks
and benefits could be managed and discussed on an individual level between health professionals and
their patients. They based this judgement on other data and the use of the vaccine overseas.
248 Cabinet Minute, CAB-21-MIN-0012, Pfizer recommendations for decision to use (9 February 2021),
https://www.health.govt.nz/system/files/2021-05/cab-21-min-0012_minute.pdf
249 Cabinet Minute, CAB-21-MIN-0322, Decision to Use the COVID-19 Pfizer Vaccine for Children Aged 12
to 15 Years (16 August 2021), https://www.health.govt.nz/system/files/2022-04/cabinet-minute-decision
to-use-the-covid-19-pfizer-vaccine-for-children-aged-12-to-15-years.pdf
250 Cabinet Minute, CAB-21-MIN-0557, COVID-19 Pfizer Vaccine for Children Aged 5 to 11 Years: Decision
to Use (20 December 2021)
251 COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use the Pfizer mRNA COVID-19 vaccine
for children aged 12 -15 years: COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations
(24 June 2021);
COVID-19 Vaccine Technical Advisory Group, Memo: Myocarditis following vaccination: COVID-19
Vaccine Technical Advisory Group (CV TAG) recommendations on the use of the Pfizer vaccine
(21 July 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19
Vaccine-Technical-Advisory-Group-CV-TAG/Myocarditis-following-vaccination.pdf;
COVID-19 Vaccine Technical Advisory Group, Memo: Priority groups for vaccination among 12- to 15- year
olds: COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations on the use of the Pfizer
vaccine (4 August 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/
COVID-19-Vaccine-Technical-Advisory-Group-CV-TAG/Priority-groups-for-vaccination-among-12-to-15
year-olds.pdf;
Ministry of Health, Memo: Extending age groups who can receive COVID-19 vaccine (13 August 2021),
https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine
Technical-Advisory-Group-CV-TAG/Extending-age-groups-who-can-receive-COVID-19-vaccine.pdf;
Cabinet Paper, CAB-21-MIN-0322, Decision to Use the COVID-19 Pfizer Vaccine for Children Aged 12 to 15
Years (16 August 2021), https://www.health.govt.nz/system/files/2022-04/cabinetpaper-decision-to-use
the-covid-19-pfizer-vaccine-for-children-aged-12-to-15-years.pdf;
Cabinet Minute, Recommendations for the Decision to Extend Use of the COVID-19 Pfizer Vaccine for
People Aged 12 to 15 Years (28 June 2021);
Ministry of Health, COVID-19 Testing Technical Advisory Group Minutes (14 December 2021);
Ministry of Health, Child Wellbeing Impact Assessment: COVID-19 immunisation for children 5 to 11 years
(20 December 2021);
COVID-19 Vaccine Technical Advisory Group, Memo: Use of the paediatric Pfizer COVID-19 vaccine in
5–11 year-olds – second dose and dosing interval: COVID-19 Vaccine Technical Advisory Group (CV TAG)
recommendations (16 February 2022), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/
Expert-groups/COVID-19-Vaccine-Technical-Advisory-Group-CV-TAG/Use-of-the-paediatric-Pfizer-COVID
19-vaccine-in-5-11-year-olds.pdf;
Cabinet Paper, CAB-21-MIN-0557, COVID-19 Pfizer Vaccine for Children Aged 5 to 11 Years: Decision to
Use (20 December 2021)
252 Cabinet Paper, CAB-21-MIN-0557, COVID-19 Pfizer Vaccine for Children Aged 5 to 11 Years: Decision to
Use (20 December 2021
253 Cabinet Social Wellbeing Committee Minute, SWC-21-MIN-0138, COVID-19 Strategy: Janssen Decision to
Use (22 September 2021)
Cabinet Minute, CAB-21-MIN-0460, AstraZeneca Vaccine: Decision to Use (8 November 2021)
Cabinet Paper and Minute, CAB-22-MIN-0046, Decision to Use Novavax’s COVID-19 Vaccine (Primary
Course) (28 February 2021), https://www.health.govt.nz/system/files/2023-08/cabinet_material_decision_
to_use_novavaxs_covid-19_vaccine_primary_course_final.pdf
254 Cabinet Paper, SWC-21-MIN-0138, COVID-19 Strategy: Janssen Decision to Use (22 September 2021)
Cabinet Paper, CAB-21-MIN-0460, AstraZeneca Vaccine: Decision to Use (8 November 2021)
Cabinet Paper and Minute, CAB-22-MIN-0046, Decision to Use Novavax’s COVID-19 Vaccine (Primary
Course) (28 February 2021), https://www.health.govt.nz/system/files/2023-08/cabinet_material_decision_
to_use_novavaxs_covid-19_vaccine_primary_course_final.pdf
255 Janssen vaccines were expected to arrive in New Zealand in early 2022. In the meantime, New Zealand
could source Vaxzevria vaccines from Australia. Cabinet Paper, CAB-21-MIN-0460, AstraZeneca Vaccine:
Decision to Use (8 November 2021)
256 Ministry of Health, Memo: Decision to use the AstraZeneca COVID-19 vaccine as a booster for those aged
18 years and over 3 months after a primary vaccine course (10 February 2022), https://www.tewhatuora.
govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine-Technical-Advisory-Group-CV
TAG/Decision-to-use-the-AstraZeneca-COVID-19-vaccine-as-a-booster-for-those-aged-18-years.pdf, p 2
257 In particular, the advice noted the risks of thrombosis and thrombocytopenia, capillary leak syndrome
and Guillain-Barré syndrome associated with Janssen and Vaxzevria vaccines.
COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use the AstraZeneca COVID-19 vaccine:
COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations (27 October 2021), https://www.
tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine-Technical-Advisory
Group-CV-TAG/27-October-2021-Decision-to-use-the-AstraZeneca-COVID-19-vaccine.pdf
COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use the Janssen COVID-19 vaccine:
COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations (12 July 2021), https://www.
tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine-Technical-Advisory
Group-CV-TAG/Decision-to-use-the-Janssen-COVID-19-vaccine.pdf
COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use a primary course of the Novavax
COVID-19 vaccine: COVID19 Vaccine Technical Advisory Group (CV TAG) recommendations (10 February
2021), https://fyi.org.nz/request/18521/response/71274/attach/5/CV%20TAG%20recommendations%20
for%20Decision%20to%20Use%20Novavax.pdf
258 Cabinet Paper, SWC-21-MIN-0138, COVID-19 Strategy: Janssen Decision to Use (22 September 2021)
259 Cabinet Paper and Minute, CAB-22-MIN-0046, Decision to Use Novavax’s COVID-19 Vaccine (Primary
Course) (28 February 2021), https://www.health.govt.nz/system/files/2023-08/cabinet_material_decision_
to_use_novavaxs_covid-19_vaccine_primary_course_final.pdf
260 Cabinet Paper, SWC-21-MIN-0138, COVID-19 Strategy: Janssen Decision to Use (22 September 2021)
261 Health New Zealand | Te Whatu Ora, Final report of the COVID-19 Vaccine Independent safety monitoring
board (3 July 2023), https://www.tewhatuora.govt.nz/publications/final-report-of-the-covid-19-vaccine
independent-safety-monitoring-board-cv-ismb
262 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, engagement with Professor
David Murdoch (5 June 2025)
NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, engagement with Vaccine
Alliance (13 May 2025)
263 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2
Section 8.4, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part
two/8-4-what-happened-vaccination-requirements
264 Cabinet Paper, COVID-19 Vaccine Strategy (June 2020), https://covid19.govt.nz/assets/Proactive-Releases/proactive-release-2020-june/PAPER-COVID-19-Vaccine-Strategy.pdf, p 10
265 Myocarditis is inflammation of the heart muscle, usually caused by an infection. Symptoms can range
from mild to severe and include chest pain, abnormal heartbeat and shortness of breath. A severe case
of myocarditis can weaken the heart, which can lead to heart failure, abnormal heartbeat and death. See
Mayo Clinic, ‘Myocarditis’ (4 June 2024), https://www.mayoclinic.org/diseases-conditions/myocarditis/symptoms-causes/syc-20352539
No vaccines were removed from the Immunisation Programme or the market by the Government
once introduced; they were only withdrawn by the companies on the basis of commercial, rather than
safety, considerations.
RNZ, ‘AstraZeneca says it will withdraw Covid-19 vaccine globally as demand dips’ (8 May 2024),
https://www.rnz.co.nz/news/world/516291/astrazeneca-says-it-will-withdraw-covid-19-vaccine-globally
as-demand-dips
Health New Zealand | Te Whatu Ora, ‘COVID-19 vaccines in Aotearoa: Novavax vaccines’ (updated
11 December 2025), https://info.health.nz/health-topics/immunisations/vaccines-aotearoa/covid-19
vaccines/covid-19-vaccines-aotearoa
266 World Health Organization, ‘The World Health Organization Programme for International Drug
Monitoring’, https://www.who.int/teams/regulation-prequalification/regulation-and-safety/
pharmacovigilance/networks/pidm
267 Uppsala Monitoring Centre, ‘About Uppsala Monitoring Centre’, https://who-umc.org/about-uppsala
monitoring-centre
268 World Health Organization, ‘The Global Advisory Committee on Vaccine Safety’, https://www.who.int/
groups/global-advisory-committee-on-vaccine-safety/
269 The Global Fund and World Health Organization, Minimum requirements for a functional
pharmacovigilance system (4 June 2010), https://cdn.who.int/media/docs/default-source/medicines/
pharmacovigilance/pv_minimum_requirements_2010_2.pdf?sfvrsn=8bfdc8, p 2
270 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
Medsafe, response to OIA request H202106950 (13 August 2021), https://www.health.govt.nz/system/
files/2021-10/h202106950_-_response.pdf
271 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
272 Medicines Act 1981, ss 24, 35–36 and 41, https://www.legislation.govt.nz/act/public/1981/0118/latest/whole.html#DLM55443
273 Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand, ed 2.2 (August 2020),
Part 8: Pharmacovigilance
Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand: Pharmacovigilance, ed
3.0 (March 2024), https://www.medsafe.govt.nz/Regulatory/Guideline/GRTPNZ/part-8-pharmacovigilance
Edition3March2024.pdf
274 The Centre for Adverse Reactions Monitoring is located within the New Zealand Pharmacovigilance
Centre at the University of Otago. In addition to contracted activities through the Centre for Adverse
Reactions Monitoring, the Pharmacovigilance Centre undertakes other work that contributes to the
safer use of therapeutic products. Michael Tatley and others, A COVID-19 Vaccine Safety Surveillance Strategy for New Zealand:
Framework Proposal for Discussion, New Zealand Pharmacovigilance Centre – University of Otago
(28 September 2020)
275 Michael Tatley and others, A COVID-19 Vaccine Safety Surveillance Strategy for New Zealand:
Framework Proposal for Discussion, New Zealand Pharmacovigilance Centre – University of Otago
(28 September 2020)
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (10 September 2025)
276 Michael Tatley and others, A COVID-19 Vaccine Safety Surveillance Strategy for New Zealand:
Framework Proposal for Discussion, New Zealand Pharmacovigilance Centre – University of Otago
(28 September 2020)
Medsafe, ‘How Does Medsafe Monitor Medicine Safety?’ (revised 21 November 2023), https://www.medsafe.govt.nz/consumers/Safety-of-Medicines/Medicines-Safety-and-Pharmacovigilance.asp
277 Medsafe, SOP-CR-02-02, Standard Operating Procedure: Signal management for adverse reactions
to medicines (30 October 2020)
278 Desiree L. Kunac, Mira Harrison-Woolrych and Michael V. Tatley, ‘Pharmacovigilance in New Zealand:
the role of the New Zealand Pharmacovigilance Centre in facilitating safer medicines use’, New Zealand
Medical Journal, vol 121:1283 (3 October 2008), https://pubmed.ncbi.nlm.nih.gov/18841188/, p 81
279 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
280 European Medicines Agency, Clinical Safety Data Management: Definitions and Standards for Expedited
Reporting (June 1995), https://www.ema.europa.eu/en/documents/scientific-guideline/international
conference-harmonisation-technical-requirements-registration-pharmaceuticals-human-use-topic-e-2
clinical-safety-data-management-definitions-and-standards-expedited-reporting-step_en.pdf, p 4
281 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
282 The Centre for Adverse Reactions Monitoring uses the World Health Organization causality assessment
criteria for this evaluation.
University of Otago, ‘New Zealand Pharmacovigilance Centre and Centre for Adverse Reactions
Monitoring’, https://nzphvc-carm.otago.ac.nz/home/
Uppsala Monitoring Centre, The use of the WHO-UMC system for standardised case causality
assessment, https://www.who.int/docs/default-source/medicines/pharmacovigilance/whocausality
assessment.pdf
283 Medsafe, ‘How does Medsafe monitor vaccine safety?’ (revised 21 November 2023), https://www.
medsafe.govt.nz/Consumers/Safety-of-Medicines/Vaccine-safety.asp
284 Steven Black, Juhani Eskola and others, ‘Importance of background rates of disease in assessment
of vaccine safety during mass immunisation with pandemic H1N1 influenza vaccine’, The Lancet,
Vol 374:9707 (19 December 2009), https://doi.org/10.1016/s0140-6736(09)61877-8
285 In practice, consent for medicines is rarely revoked. Medsafe, ‘How does Medsafe monitor medicine
safety?’ (revised 21 November 2023), https://www.medsafe.govt.nz/Consumers/Safety-of-Medicines/
Medicines-Safety-and-Pharmacovigilance.asp
286 Medsafe, ‘Medsafe Safety Communications’ (revised 15 December 2025), https://www.medsafe.govt.nz/
safety/SafetyCommunications.asp
287 Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand, ed 2.2 (August 2020),
Part 8: Pharmacovigilance
Medsafe, Guideline on the Regulation of Therapeutic Products in New Zealand: Pharmacovigilance, ed
3.0 (March 2024), https://www.medsafe.govt.nz/Regulatory/Guideline/GRTPNZ/part-8-pharmacovigilance
Edition3March2024.pdf
288 Cabinet Paper, COVID-19 Vaccine Strategy (June 2020), https://covid19.govt.nz/assets/Proactive-Releases/proactive-release-2020-june/PAPER-COVID-19-Vaccine-Strategy.pdf, p 10
289 Michael Tatley and others, A COVID-19 Vaccine Safety Surveillance Strategy for New Zealand:
Framework Proposal for Discussion, New Zealand Pharmacovigilance Centre – University of Otago
(28 September 2020)
Ministry of Health, COVID-19 Vaccine Safety Monitoring (November 2020)
Medsafe, Vaccine safety surveillance for COVID-19 vaccines – Medsafe planning proposal [n.d]
290 Michael Tatley and others, A COVID-19 Vaccine Safety Surveillance Strategy for New Zealand:
Framework Proposal for Discussion, New Zealand Pharmacovigilance Centre – University of Otago
(28 September 2020)
291 Medsafe, Vaccine safety surveillance for COVID-19 vaccines – Medsafe planning proposal [n.d]
292 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
293 See section 2.1.2.5 for an explanation of Risk Management Plans.
294 The groups provided advice and recommendations, but did not have decision-making responsibilities,
powers or functions.
295 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
296 Both groups are technical advisory committees established under section 8 of the Medicines Act 1981
to advise Medsafe and the Minister of Health. The Medicines Assessment Advisory Group advises on the
risk-benefit profile of new medicines, and the Medicines Adverse Reactions Committee advises on the
safety of approved medicines.
Medsafe, ‘Committees’ (revised 17 March 2021), https://www.medsafe.govt.nz/committees/committees
landing.asp
297 Meetings were usually held fortnightly but were more frequent during high demand periods and
became monthly towards the end of the Group’s existence.
298 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
299 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
300 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
301 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
302 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022
303 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
304 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025)
305 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
306 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
307 The independent Best Practice Advocacy Centre New Zealand also published a short article for
healthcare providers explaining how to make a report to the Centre for Adverse Reactions Monitoring.
Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025);
Medsafe, ‘New reporting form for adverse events following immunisation with COVID-19 vaccines’
(4 March 2021), https://www.medsafe.govt.nz/profs/PUArticles/March2021/New-reporting-form-for-AEFI
with-COVID-19-vaccines.html
Medsafe, Vaccine Adverse Event Reporting Form (revised 19 December 2022), https://www.medsafe.govt.
nz/COVID-19/adverse-event-reporting-form.asp
Best Practice Advocacy Centre New Zealand, Best Practice Bulletin: Issue 19 (23 February 2021),
https://bpac.org.nz/bulletin/bestpractice/nineteen.aspx
308 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
309 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
310 Office of the Chief Coroner, written response to NZ Royal Commission of Inquiry into COVID-19 Lessons
Learned: Phase Two request for information (15 August 2025)
311 Ministry of Health, Brief of Evidence of Christine May Nolan (19 August 2022)
312 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
313 Medsafe, Vaccine safety surveillance for COVID-19 vaccines – Medsafe planning proposal, n.d
314 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
315 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025)
316 Medsafe, Vaccine safety surveillance for COVID-19 vaccines – Medsafe planning proposal, n.d
317 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
318 A memorandum requesting funding for this research was provided to the Director-General of Health
on 15 January 2021, but a signed version of that memorandum was unable to be located.
319 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 March 2025)
320 Although the results were not published until March 2022, the Ministry of Health said it had access to
estimates of background rates of adverse event before then, and that the results from the SAFE study
provided an additional dataset.
Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025)
321 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
322 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
323 The New Zealand Immunisation Handbook provides clinical guidelines for health professionals on the
safest and most effective use of vaccines in their practice; they are used widely in general practice.
Health New Zealand | Te Whatu Ora, Immunisation Handbook 2025, version 5 (10 September 2025),
https://www.tewhatuora.govt.nz/for-health-professionals/clinical-guidance/immunisation-handbook
324 Accident Compensation Corporation, response to production order issued by NZ Royal Commission
of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013
(03 April 2025)
325 ACC has a legal obligation to notify relevant authorities if it believes, based on information collected
while processing claims, that there is a risk of harm to the public. Accident Compensation Act 2001,
s 284, https://www.legislation.govt.nz/act/public/2001/0049/latest/DLM103407.html
Accident Compensation Corporation, response to production order issued by NZ Royal Commission
of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013
(03 April 2025)
326 Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
327 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, email to the Health and
Disability Commissioner (29 July 2025)
Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability
Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf
328 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two, email to the Health Quality
and Safety Commission (11 August 2025)
329 Pericarditis is inflammation of the sac around the heart. Symptoms are very similar to myocarditis and
pericarditis may often occur with myocarditis. Mayo Clinic, ‘Pericarditis’ (9 April 2024), https://www.
mayoclinic.org/diseases-conditions/pericarditis/symptoms-causes/syc-20352510
Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
Ministry of Health, Affidavit of Christopher Mark James, CIV-2022-485-13 (21 January 2022)
330 Navya Voleti, Surya Prakash Reddy and Paddy Ssentongo, ‘Myocarditis in SARS-CoV-2 infection vs
COVID-19 vaccination: A systematic review and meta-analysis’, Frontiers in Cardiovascular Medicine,
Vol 9 (29 August 2022), https://doi.org/10.3389/fcvm.2022.951314
NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two (2026), Pandemic
Perspectives, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/phase-two/
pandemic-perspectives
331 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
332 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
333 COVID-19 Vaccine Independent Safety Monitoring Board, Minutes (29 April 2021)
Ministry of Health, response to Production Order issued by NZ Royal Commission of Inquiry into
COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (15 August 2025)
Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
334 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
335 Safety reports provide an overview of the reported adverse events following immunisation in
New Zealand, and any potential safety concerns under investigation. The reports were published
weekly on Medsafe’s website for most of 2021, but are usually bi-monthly. The reports also contain links
to information produced by Medsafe. Ministry of Health, Brief of Evidence of Dr Timothy Robert George
Hanlon (19 August 2022)
336 United States Centers for Disease Control and Prevention, COVID-19 VaST Work Group Report – May 17,
2021 (reviewed 20 May 2021), https://archive.cdc.gov/www_cdc_gov/vaccines/acip/work-groups-vast/
report-2021-05-17.html
World Health Organization, ‘COVID-19 subcommittee of the WHO Global Advisory Committee on Vaccine
Safety (GACVS) reviews cases of mild myocarditis reported with COVID-19 mRNA vaccines’ (26 May 2021),
https://www.who.int/news/item/26-05-2021-gacvs-myocarditis-reported-with-covid-19-mrna-vaccines
337 COVID-19 Vaccine Independent Safety Monitoring Board, Minutes (27 May 2021)
Medsafe, Memo: Comirnaty and potential myocarditis (24 May 2021), https://www.health.govt.nz/system/
files/2022-08/h202201068_-_response.pdf
338 A monitoring communication provides information to consumers and healthcare professionals about
newly-identified potential safety concerns. The monitoring period was to end on 31 December 2021.
Medsafe, ‘Myocarditis – a potential adverse reaction to Comirnaty (Pfizer COVID-19 vaccine)’ (9 June 2021),
https://www.medsafe.govt.nz/safety/Alerts/comirnaty-myocarditis.asp
Medsafe, ‘Medsafe Safety Communications’ (revised 15 December 2025), https://www.medsafe.govt.nz/
safety/SafetyCommunications.asp
339 The COVID-19 Vaccine Update was a fortnightly newsletter to the health and disability sector, sent
to about 15,000 subscribers and immunisation programme contacts.
Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
340 New Zealand Gazette, 2021-go2440, Provisional Consent to the Distribution of a New Medicine
(21 June 2021), https://gazette.govt.nz/notice/id/2021-go2440
341 Cabinet Minute, Recommendations for the Decision to Extend Use of the COVID-19 Pfizer Vaccine for
People Aged 12 to 15 Years (28 June 2021)
342 COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use the Pfizer mRNA COVID-19 vaccine
for children aged 12 – 15 years: COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations
(24 June 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID
19-Vaccine-Technical-Advisory-Group-CV-TAG/Decision-to-use-the-Pfizer-mRNA-COVID-19-vaccine-for
children-aged-12-15.pdf
343 Data from the United States suggested that – in 12–29 year olds – the incidence of myocarditis following
vaccination was around 40.6 cases per million second doses in males, and 4.2 cases per million in
females. For adults aged 30 or older the incidence per million was 2.4 in men and 1.0 in women.
World Health Organization, COVID-19 subcommittee of the WHO Global Advisory Committee on
Vaccine Safety (GACVS): updated guidance regarding myocarditis and pericarditis reported with
COVID-19 mRNA vaccines (9 July 2021), https://www.who.int/news/item/09-07-2021-gacvs-guidance
myocarditis-pericarditis-covid-19-mrna-vaccines
344 COVID-19 Vaccine Independent Safety Monitoring Board, Minute (24 June 2021)
345 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
346 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
COVID-19 Vaccine Technical Advisory Group, Memo: Decision to use the Pfizer mRNA COVID-19 vaccine
for children aged 12–15 years: COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations
(24 June 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID
19-Vaccine-Technical-Advisory-Group-CV-TAG/Decision-to-use-the-Pfizer-mRNA-COVID-19-vaccine-for
children-aged-12-15.pdf
COVID-19 Vaccine Technical Advisory Group, Memo: Myocarditis following vaccination: COVID-19
Vaccine Technical Advisory Group (CV TAG) recommendations on the use of the Pfizer vaccine
(21 July 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19
Vaccine-Technical-Advisory-Group-CV-TAG/Myocarditis-following-vaccination.pdf
347 Medsafe, ‘Myocarditis and pericarditis – rare adverse reactions to Comirnaty (Pfizer COVID-19 vaccine)’
(21 July 2021), https://www.medsafe.govt.nz/safety/Alerts/comirnaty-myocarditis-alert.htm
348 The communication advised ‘[i]t has now been concluded by Medsafe, other national regulators (such as
the Food and Drug Administration) and the company, Pfizer, that myocarditis (inflammation of the heart
muscle) is a rare side effect of vaccination with Comirnaty.’
Medsafe, ‘Myocarditis and pericarditis – rare adverse reactions to Comirnaty (Pfizer COVID-19 vaccine)’
(21 July 2021), https://www.medsafe.govt.nz/safety/Alerts/comirnaty-myocarditis-alert.htm
349 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
350 Ministry of Health, Datasheet: Comirnaty COVID-19 Vaccine (2 August 2021)
Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
351 Ministry of Health, Brief of Evidence of Christopher Mark James (18 August 2021)
352 Ministry of Health, Brief of Evidence of Christine May Nolan (19 August 2022)
353 COVID-19 Vaccine Technical Advisory Group, Memo: Myocarditis following vaccination:
COVID-19 Vaccine Technical Advisory Group (CV TAG) recommendations on the use of the Pfizer
vaccine (21 July 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/
COVID-19-Vaccine-Technical-Advisory-Group-CV-TAG/Myocarditis-following-vaccination.pdf
354 Compared to the standard 21-day interval at the time.
355 Ministry of Health, HR202111680, Recommendations from the COVID-19 Vaccine Technical Advisory
Group on the risk of myocarditis and pericarditis following vaccination (22 July 2021)
356 Ministry of Health, COVID-19 Vaccine Technical Advisory Group, Minutes (27 July 2021)
357 COVID-19 Vaccine Technical Advisory Group, Minutes (3 August 2021)
358 Ministry of Health, COVID-19 Vaccine Technical Advisory Group, Agenda and Supporting Papers (27 July 2021)
359 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
360 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
361 Rt. Hon. Jacinda Ardern, ‘Government sets out plan to reconnect New Zealanders to the world’ (12 August 2021), https://www.beehive.govt.nz/release/government-sets-out-plan-reconnect-newzealanders-world
362 Dan Satherley, ‘Coronavirus: Gap between Pfizer COVID-19 vaccine doses extended to six weeks’, Stuff (21 August 2021), https://www.stuff.co.nz/nz-news/350519563/coronavirus-gap-between-pfizer
covid-19-vaccine-doses-extended-to-six-weeks
363 COVID-19 Vaccine Technical Advisory Group, Memo: Extending age groups who can receive COVID-19 vaccine (13 August 2021), https://www.tewhatuora.govt.nz/assets/About-us/Who-we-are/Expert-groups/COVID-19-Vaccine-Technical-Advisory-Group-CV-TAG/Extending-age-groups-who-can-receive-COVID-19vaccine.pdf Cabinet minute, CAB-21-MIN-0322, Decision to Use the COVID-19 Pfizer Vaccine for Children Aged 12 to 15 Years (16 August 2021), https://www.health.govt.nz/system/files/2022-04/cabinet-minute-decision-touse-the-covid-19-pfizer-vaccine-for-children-aged-12-to-15-years.pdf
364 Cabinet Paper, CAB-21-MIN-0322, Decision to Use the COVID-19 Pfizer Vaccine for Children Aged 12 to 15 Years (16 August 2021), https://www.health.govt.nz/system/files/2022-04/cabinetpaper-decision-to-usethe-covid-19-pfizer-vaccine-for-children-aged-12-to-15-years.pdf
365 Cabinet Minute, CAB-21-MIN-0324, COVID-19: Auckland Community Case (17 August 2021), https://www.dpmc.govt.nz/sites/default/files/2023-01/ALC1-17082021-Auckland-Community-Case.pdf Press conference, Hansard transcript (17 August 2021), https://www.beehive.govt.nz/sites/default/files/2021-08/Press%20conference%2017%20August%202021.pdf
366 Cabinet Minute, CAB-21-MIN-0557, COVID-19 Pfizer Vaccine for Children Aged 5 to 11 Years: Decision to Use (20 December 2021)
367 Rt. Hon. Chris Hipkins, ‘Government confirms COVID-19 vaccinations to protect tamariki’ (21 December 2021), https://www.beehive.govt.nz/release/government-confirms-covid-19-vaccinations
protect-tamariki
368 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022) Ministry of Health, Brief of Evidence of Christine May Nolan (19 August 2022)
369 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022) Ministry of Health, Brief of Evidence of Christine May Nolan (19 August 2022)
370 Ministry of Health, Brief of Evidence of Dr Timothy Robert George Hanlon (19 August 2022)
371 Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf, p 19
372 Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf, pp 6–7
373 Ministry of Health, COVID-19 Vaccine Independent Safety Monitoring Board, Minutes (22 June 2022) Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025)
374 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025)
375 COVID-19 Vaccine Independent Safety Monitoring Board, Final report 2022: COVID-19 Vaccine Independent Safety Monitoring Board (3 July 2023), https://www.tewhatuora.govt.nz/publications/finalreport-of-the-covid-19-vaccine-independent-safety-monitoring-board-cv-ismb
376 The Ministry of Health commissioned new research to help it identify safety signals resulting from reports of adverse events, relying on a scientifically established understanding of the background rates of adverse events in the population (see section 2.1.6.8).
377 While reports were made in respect of the AstraZeneca and Janssen vaccines, those reports did not result in safety signal investigations. This is likely because the adverse reactions were known at the time of approval, as discussed in section 2.1.2.7. COVID-19 Vaccine Independent Safety Monitoring Board, Final report 2022: COVID-19 Vaccine Independent Safety Monitoring Board (3 July 2023), https://www.tewhatuora.govt.nz/publications/finalreport-of-the-covid-19-vaccine-independent-safety-monitoring-board-cv-ismb, p 11
378 Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf, p 19
379 Ministry of Health, response to production order issued by NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase Two under section 20 of the Inquiries Act 2013 (7 October 2025) Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf, p 6
380 Health and Disability Commissioner, Case 22HDC02256, A Report by the Health and Disability Commissioner (9 April 2024), https://www.hdc.org.nz/media/wmubfvcz/22hdc02256.pdf, p 19
381 The Code of Health and Disability Services Consumers’ Rights places the duty on the health and disability service providers to inform consumers of risks and obtain informed consent before providing services. Health and Disability Commissioner (Code of Health and Disability Services Consumers’ Rights) Regulations 1996, Schedule, https://www.legislation.govt.nz/regulation/public/1996/0078/latest/
DLM209085.html
382 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2 Section 7.3.1.3, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/
part-two/7-3-our-assessment
383 NZ Royal Commission of Inquiry into COVID-19 Lessons Learned: Phase One, Main Report (2024), Part 2 Section 7.3.1.3, https://www.covid19lessons.royalcommission.nz/reports-lessons-learned/main-report/part-two/7-3-our-assessment

